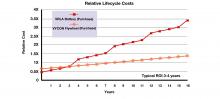
Kona Medical announced three and six month results from the WAVE I study, a first-in-man study evaluating the safety and efficacy of Kona Medical’s Surround Sound Renal Denervation System for the treatment of resistant hypertension.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now