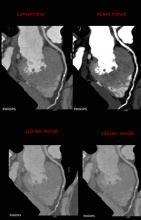
November 16, 2015 — At the Radiological Society of North America (RSNA) 2015 annual meeting, GE Healthcare will unveil the next generation of its Logiq E9 general imaging ultrasound system. The system combines the power of XDclear probes with a new XDclear 2.0 platform that maximizes spatial and contrast resolution. As a result, XDclear 2.0 optimizes the imaging from the pulse of the probe to each of its pixels and every step in between.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now