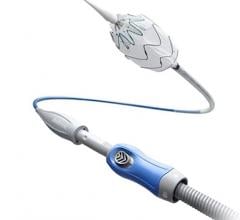
December 29, 2010 – The U.S. Food and Drug Administration (FDA) has approved a stent graft system for treating abdominal aortic aneurysms (AAA). The Endurant AAA Stent Graft System, from Medtronic, can be used for the minimally invasive treatment of the often fatal condition.
An estimated 1.2 million people in the United States have an AAA – a dangerous and often asymptomatic bulge or ballooning in their bodies’ main artery – and most of them don’t know it. The new system is a safe and effective treatment option that doesn’t require major surgery, an extended hospital stay or a long recovery period.
It is a flexible wire frame (stent) sewn onto a specially woven fabric tube (graft) that physicians use to create a new path for blood flow in the patient’s aorta. This reduces pressure on the aneurysm and the risk of rupture. The new device is designed to conform to a broad range of aortic anatomies and is delivered through catheters inserted into blood vessels in the groin.
“With FDA approval of Medtronic’s Endurant stent graft, U.S. physicians now have access to a new-generation device that will allow safe treatment of even more complex AAA patients with EVAR than was feasible with previous devices,” said Michel Makaroun, M.D., professor and chief of vascular surgery at the University of Pittsburgh Medical Center, and the principal investigator for the U.S. clinical study. “With its innovative design and delivery system, the Endurant stent graft represents another significant advance that expands our toolkit for the minimally invasive management of this dangerous, often deadly, condition.”
In the study, there were no post-implant aneurysm ruptures or aneurysm-related deaths through one year of patient follow-up, and no patients experienced enlargement of their aneurysms during this timeframe. All remained stable or shrank in size. Also through one year, there were no device migrations or device-related “endoleaks,” which can result in persistent blood flow into the aneurysm sac and enlargement of the aneurysm. The study involved 150 patients and 26 medical centers, and successfully met its primary goals and endpoints for assessing the safety and effectiveness of the Endurant stent graft.
AAAs rarely cause symptoms until they rupture, which usually results in death due to rapid and extensive internal bleeding. They are often found coincidentally on X-rays or other medical imaging studies performed for other reasons. Detection and diagnosis of this condition are critical to its effective management. The Society for Vascular Surgery recommends treatment for AAAs that are 5.5 cm or greater in diameter.
Physicians rely primarily on two approaches to treat AAAs: open surgical repair and endovascular repair. Open surgical repair requires a large incision in the abdomen to access the aorta. It involves removing the aneurysmal segment and replacement with a synthetic graft that is sewn onto healthy aortic tissue. Endovascular repair is a less invasive procedure in which a stent graft – a wire mesh supporting a fabric tube – is compressed on a delivery catheter, threaded through an artery in the groin and expanded at the site of the aneurysm. It does not require removal of the aneurysmal segment.
For more information: www.medtronic.com


 September 18, 2025
September 18, 2025