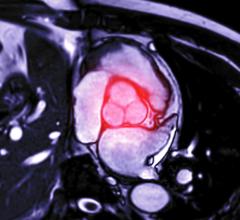
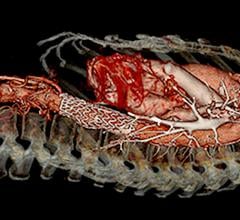
May 12, 2021 — Preliminary results of a clinical trial, presented today at the AATS 101st Annual Meeting, showed that a new, low-profile thoracic aortic endograft is safe and effective in the treatment of descending thoracic aortic aneurysm or penetrating atherosclerotic ulcer (PAU) diseases. A multi-disciplinary team, led by both cardiac and vascular surgeons as co-investigators, conducted the study in 36 centers in the United States and Japan, enrolling patients between 2016 and 2019.
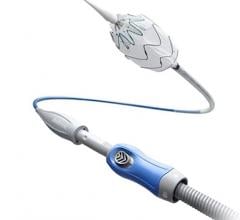
The trial aimed to measure safety and efficacy of the RELAYPro endovascular device, a second-generation product featuring a dramatically reduced profile and a non-bare stent (NBS) configuration.
The prospective, international, non-blinded, non-randomized pivotal trial analyzed a primary safety endpoint of major adverse events (MAE) at 30 days (death, myocardial infarction, stroke, renal/respiratory failure, paralysis, bowel ischemia, procedural blood loss) and a primary effectiveness endpoint of treatment success at one year (technical success, patency, absence of aneurysm rupture, type I/III endoleaks, stent fractures, secondary interventions, aneurysm expansion, and migration). Treatment success at one year was 89.2 percent.
“With a 3 to 4 French profile reduction, this second generation thoracic endograft device met the one year safety and effectiveness endpoints in a pivotal study for the treatment of patients with aneurysms of the descending thoracic aorta or PAUs,” explained Wilson Szeto, M.D., Professor of Surgery at the Hospital of the University of Pennsylvania and Penn Presbyterian Medical Center. “It is particularly positive to note that the reduction in profile and the availability of a non bare stents configuration will expand the population of patients who can be treated with the device and reduces complications.”
In this study, the vast majority of patients in the United States were treated with a percutaneous approach, which can dramatically reduce surgical complications associated with higher profile devices requiring surgical cutdown for deployment. At one year follow up, patients demonstrated a low risk of mortality, endoleak or structural integrity concerns. Follow-up continues to five years and the device is currently being evaluated for approval by the FDA.
For more information: www.aats.org


 September 18, 2025
September 18, 2025