St. Jude Medical Inc. announced CE Mark approval and launch of SyncAV CRT software, designed to…
GE Healthcare and Getinge Group announced the U.S. launch of a new, highly flexible angiography…
At the 2016 Annual Meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI),…
Vital Connect Inc., developer of wearable biosensor technologies, and BePATIENT, a startup that…
Healthy Interactions and MedCurrent recently introduced a first-of-its-kind clinical decision…
June 2, 2016 — Artec 3D, a developer and manufacturer of professional 3-D hardware and software…
Biotronik announced the launch of CardioMessenger Smart in the United States. CardioMessenger…
MIM Software Inc. announced it has received 510(k) U.S. Food and Drug Administration (FDA)…
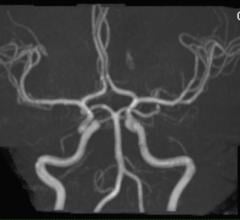
Medic Vision Imaging Solutions Ltd. announced the U.S. Food and Drug Administration (FDA)…
Boston Scientific has received U.S. Food and Drug Administration (FDA) approval for two…
May 18, 2016 — The Intersocietal Accreditation Commission (IAC) recently announced its launch of…
May 12, 2016 — CareTaker Medical announced that the U.S.
Medtronic plc recently announced it received U.S. Food and Drug Administration (FDA) approval…
Biotronik announced U.S. Food and Drug Administration (FDA) approval of Iperia ProMRI HF-T, a…
May 2, 2016 — Hitachi Medical Systems America Inc.
Bayer announced that the U.S. Food and Drug Administration (FDA) has approved Gadavist (…
April 29, 2016 — AtriCure Inc. announced U.S.
Konica Minolta introduced the latest version of the Sonimage HS1 compact ultrasound system,…
Boston Scientific has received U.S. Food and Drug Administration (FDA) approval for a suite of…
Veniti Inc. announced the first successful treatment with the Vici Verto Venous Stent System of…


 June 28, 2016
June 28, 2016