Mast Therapeutics Inc. announced that, in a placebo-controlled, nonclinical model of chronic heart failure, MST-188 demonstrated a statistically significant improvement in numerous parameters of heart function, including left ventricular ejection fraction and end-systolic volume, stroke volume and cardiac output.
Taking their pick, biomedical researchers can now conduct five different imaging studies in one scan with a state-of-the-art preclinical molecular imaging system that scientists unveiled during the Society of Nuclear Medicine and Molecular Imaging’s 2014 Annual Meeting.
June 10, 2014 — Ariad Pharmaceuticals Inc. and Medinol Ltd. earlier this year initiated two registration trials of Medinol’s NIRsupreme ridaforolimus-eluting coronary stent system incorporating Ariad’s mTOR inhibitor, ridaforolimus. The two NIRsupreme trials are randomized, single-blind, global studies taking place in the United States, Europe, Israel and Canada and will enroll approximately 2,200 patients with coronary artery disease.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
June 10, 2014 — A real-time monitoring device that beeps in the presence of high doses of radiation can help reduce patient and interventional cardiologist exposure to radiation during cardiac catheterization, according to results of the RadiCure study presented as a late-breaking clinical trial at the Society for Cardiovascular Angiography and Interventions (SCAI) 2014 scientific sessions in Las Vegas.

June 9, 2014 — After one year, patients treated with orbital atherectomy, a procedure to sand away calcium in the coronary arteries prior to stenting, were less likely to require a repeat procedure to reopen their vessel or suffer a heart attack or death compared to other published trials in patients with heavily calcified arteries. The new results from the ORBIT II study were presented as a late-breaking clinical trial at the Society for Cardiovascular Angiography and Interventions (SCAI) 2014 scientific sessions in Las Vegas.
June 9, 2014 — Kalila Medical’s Vado steerable introducer sheath for vascular and intracardiac access, including atrial fibrillation (AF) procedures, has CE mark approval.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Researchers at the Society of Nuclear Medicine and Molecular Imaging’s 2014 Annual Meeting (SNMMI) revealed how a protein encourages the production of stem cells that regenerate damaged tissues of the heart following an acute attack (myocardial infarction). They further assert that it has a better chance of working if provided early in treatment. This was confirmed by molecular imaging, which captured patients’ improved heart health after therapy.
At the 2014 Annual Meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI), June 7-11, at the St. Louis Convention Center, Siemens Healthcare will debut syngo.via for molecular imaging — a reading solution that transforms large amounts of data into comprehensive results, allowing for organ-based reading [1], reference-based quantification [2] and evidence-based reporting [3] of disease.
At the Society of Nuclear Medicine and Molecular Imaging (SNMMI) Annual Meeting, GE Healthcare introduced its Discovery* IQ^ PET/CT (positron emission tomography/ computed tomography) system enabling both outstanding image quality and intelligent quantitation, helping physicians to deliver the best possible patient outcomes.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Continuing to expand its portfolio of endovascular solutions, Medtronic Inc. announced that two recently U.S. Food and Drug Administration (FDA) approved devices will be featured at the Society for Vascular Surgery’s 2014 Vascular Annual Meeting, taking place June 5-7 in Boston.
Wolters Kluwer Health announced that Kettering Health Network has selected ProVation Order Sets, powered by UpToDate Decision Support, as its clinical content management system.
Among the collection of aspiring medical isotope production companies looking to build facilities in the United States, Coquí RadioPharmaceuticals Corp. is he only one using a design and production method already proven in the commercial markets. Coquí is planning on constructing and operating a dedicated medical isotope production facility.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
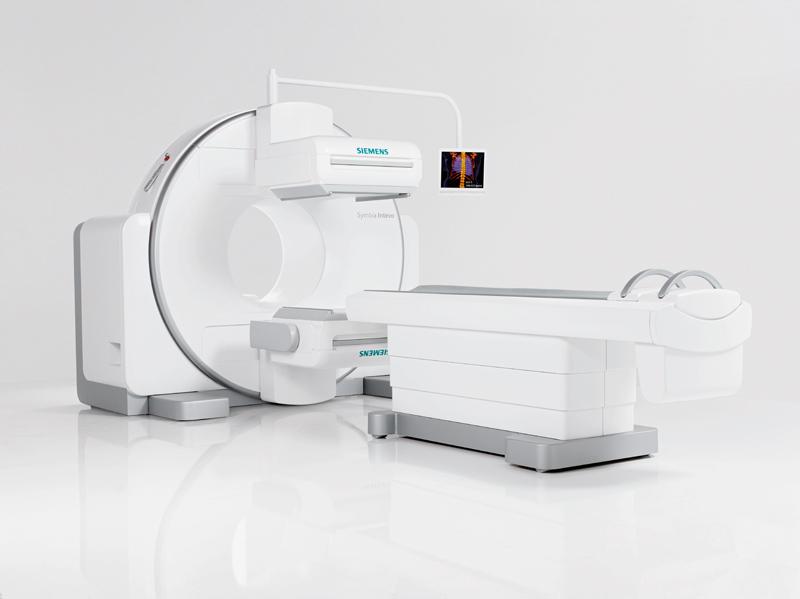
June 5, 2014 — Fairview Health Services, Minneapolis, recently became the first U.S. healthcare facility to install the Symbia Intevo xSPECT system from Siemens Healthcare. The first ever xSPECT system, the Symbia Intevo fully integrates single-photon emission computed tomography (SPECT) and CT during image reconstruction, combining SPECT’s high sensitivity with the high specificity of CT and completely integrating data from the two modalities to generate high resolution and, for the first time, quantitative images.
June 5, 2014 — Mercator MedSystems is in the midst of its prospective, 300-patient, 30-site DANCE (Dexamethasone to the Adventitia to eNhance Clinical Efficacy) clinical trial to study a new therapeutic approach to treating peripheral artery disease (PAD). The trial enrolled its first patient in November 2013, and incorporates the use of Mercator Med Systems’ U.S. Food and Drug Administration (FDA)-cleared Micro-Infusion drug delivery system.
June 5, 2014 — Lumedx Corp. and Scientific Software Solutions Inc. (SSS), maker of PedCath congenital catheterization reporting software, announced they are partnering to create an innovative solution for congenital cardiac data and image management.

 June 10, 2014
June 10, 2014








