A growing trend toward physician preference for magnetic resonance imaging (MRI) in diagnosing heart disease represents ...
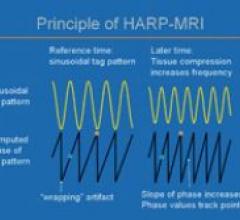
Diagnosoft HARP is software that assists physicians in the analysis of magnetic resonance (MR) images by providing ...
A low osmolar, nonionic, iodinated contrast agent, the OMNIPAQUE has been used in more than 250 million patients ...
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
Feb. 7, 2007 — Cardium Therapeutics and its operating unit InnerCool Therapies have announced they will launch two next ...
Providing clinicians with enterprise-wide data analysis of information stored in an Oracle-supported database, Misys ...
GE Healthcare has launched its new LightSpeed VCT XT, the industry’s first CT scanner that maintains high image quality ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Rapidly cooling a patient's core temperature, the ThermoSuit System consists of a disposable body suit and a means to ...
Feb. 6, 2007 — The FDA has issued an approval letter for GlaxoSmithKline’s once-a-day anticoagulant, Arixtra ...
Feb. 6, 2007 — The Centers for Medicare and Medicaid Services (CMS) has released new proposed policy in which coverage ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Feb. 1, 2007 — Cook Medical released nine-month data this week for its PTX Paclitaxel-Eluting Stent (DES) trial, which ...
Feb. 1, 2007 - New data shows that a year after undergoing carotid angioplasty and stenting, a minimally invasive ...
Feb. 1, 2007 — Stockholders of Conor Medsystems, Inc. have approved the merger agreement between Conor and Johnson & ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Feb. 1, 2007 — Royal Philips Electronics has announced the commercial release of its new EP Navigator, which Philips ...
Jan. 31, 2007 — Boston Scientific Corp. days it has received FDA 510(k) clearance to expand its PolarCath Peripheral Dilatation System offering to include a 100mm balloon.
Jan. 31, 2007 — Possis Medical, Inc., a developer, manufacturer and distributor of endovascular medical devices, announced yesterday that it has FDA approval to being marketing AngioJet Spiroflex rapid exchange catheter to remove blood clots (thrombus) from coronary arteries.

 February 06, 2007
February 06, 2007