August 11, 2010 – New sizes of the SoloPath TransFemoral Endovascular Access Catheter were recently cleared by ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
August 9, 2010 - Health care professionals will attend a conference devoted to the fundamentals of establishing ...
August 9, 2010 – Enrollment was recently completed in a clinical trial of Medtronic’s Complete SE (self-expanding) ...
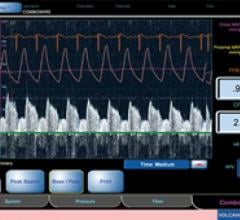
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
August 9, 2010 – According to Millennium Research Group (MRG), a medical technology market research company ...
August 4, 2010 — A second-generation of the Diamondback Predator 360 peripheral arterial disease (PAD) system ...
August 3, 2010 — Patient enrollment began in the EVOLVE clinical trial, which is designed to assess the safety and ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
August 3, 2010 — St. Joseph’s Regional Medical Center (SJRMC) in Paterson, N.J., received the Chain of Survival ...
July 30, 2010 – A drug-eluting coronary bifurcation stent received CE mark this week for clinical use in Europe ...
July 29, 2010 – A new, highly anticipated antiplatelet drug was recommended yesterday for final approval by the U ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
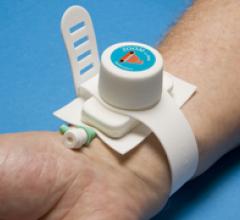
July, 29, 2010 – A second generation transradial compression bracelet is designed to obtain a stable compression ...
July 28, 2010 – This week St. Jude Medical filed a patent infringement lawsuit against Volcano Corp. over its ...
July 26, 2010 – Today a fourth-generation intracranial aneurysm stent
system was released in both the U.S. and ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
July 26, 2010 – The U.S. Food and Drug Administration (FDA) approved the first generic version of Lovenox ...

Cardiac rhythm device reliability and the approach to managing complications associated with these devices have recently ...
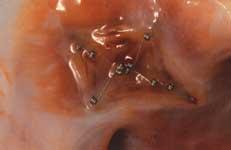
In addition to the FDA-cleared Gore Helex and the AGA Amplatzer transcatheter devices for atrial septal defects ...


 August 11, 2010
August 11, 2010