January 6, 2021 — The U.S. Food and Drug Administration (FDA) has granted the designation of breakthrough device to P+F ...
Heart Valve Technology
This channel includes news and new device innovations about heart valve technologies, including the aortic valve, mitral valve, pulmonic valve, and tricuspid valve. This includes information on transcatheter valve technologies like transcatheter aortic valve replacement (TAVR, or implantation TAVI), transcatheter mitral valve repair or replacement (TMVR), transcatheter and surgical valve repairs, and surgical replacement valves. Newer devices are now being used for transcatheter tricuspid valve repair replacement (TTVR).
December 29, 2020 — Edwards Lifesciences announced Dec. 21 that the first patient was treated in the RESTORE clinical ...
December 29, 2020 — Health Canada has approved the expanded use of the Edwards Lifesciences Sapien 3 and Sapien 3 Ultra ...
Here are the top 25 best performing articles on the Diagnostic and Interventional Cardiology (DAIC) website from 2020 ...
December 14, 2020 - Foldax Inc. today announced that the U.S. Food and Drug Administration (FDA) has granted an ...
December 1, 2020 — A recent publication demonstrated procedural efficiency for MitraClip transcatheter mitral valve ...
November 18, 2020 — The primary results from the RIVER Trial, Rivaroxaban for Valvular Heart disease and Atrial ...
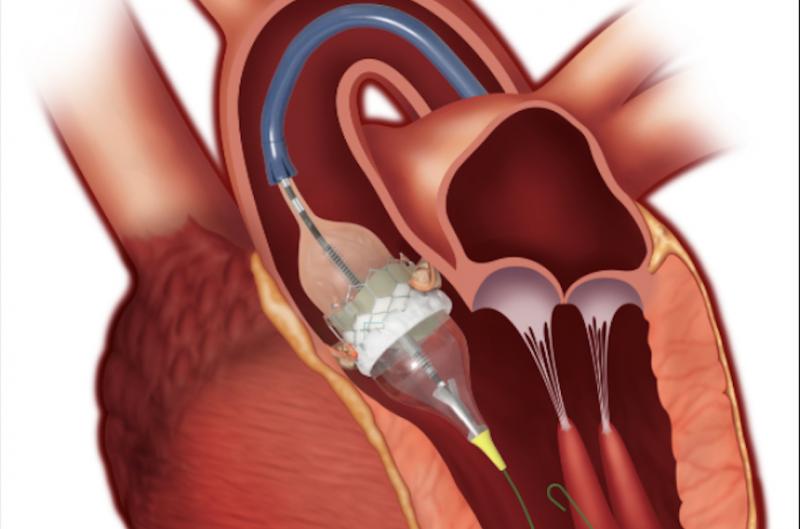
November 17, 2020 — Since the approval of the first transcatheter aortic valve replacement (TAVR) device in 2011, more ...

November 17, 2020 — Boston Scientific Corp. announced today it is immediately retiring the entire Lotus Edge ...
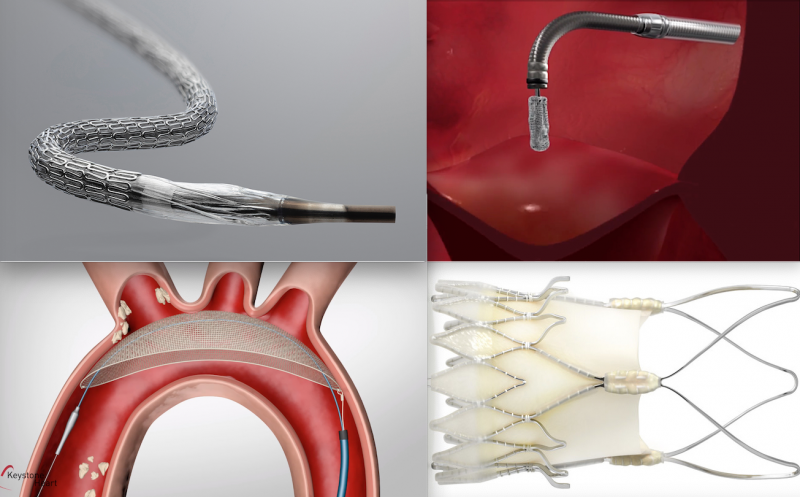
Here are some of the key takeaways from the late-breaking interventional cardiology and structural heart trials ...
David Cohen, M.D., presents late-breaking data from the STS/ACC Transcatheter Valve Registry (TVT) showing the impact of ...
Doctor Hans-Josef Feistritzerm, Heart Center of Leipzig, Germany, presents data on the use of general vs. local ...
The late-breaking MitraBridge Study was presented at Transcatheter Cardiovascular Therapeutics (TCT) 2020 meeting showed ...
October 15, 2020 – The REFLECT II randomized clinical trial evaluating the safety and efficacy of the Keystone Heart ...
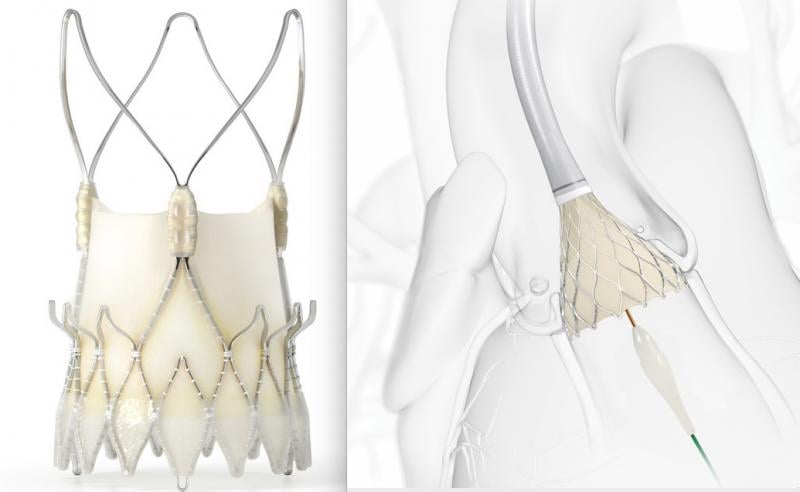
October 15, 2020 – The SCOPE II trial comparing the Boston Scientific Acurate neo vs. Medtronic CoreValve Evolut TAVR ...


 January 06, 2021
January 06, 2021