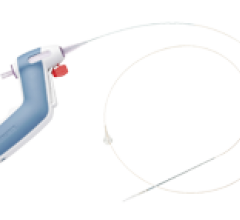
August 12, 2014 — Teleflex Inc.’s subsidiary Hotspur Technologies Inc. received U.S. Food and Drug Administration (FDA) ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
August 8, 2014 — Biotronik announced the completion of patient enrollment in the superficial femoral artery (SFA) arm of ...
July 29, 2014 — Biotronik announced that the first patient has been enrolled in the investigator-initiated BIOLUX 4EVER ...
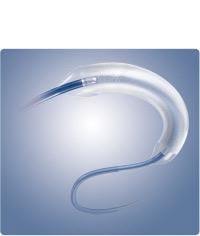
July 28, 2014 — Boston Scientific Corp. received CE mark for the Ranger Paclitaxel-coated PTA Balloon Catheter. The ...
July 18, 2014 — Avinger announced that John Pigott, M.D., of Jobst Vascular Institute, Toledo, Ohio, enrolled the first ...
July 11, 2014 — Cordis Corp. announced the launch of its Saber PTA (percutaneous transluminal angioplasty) dilatation ...

June 16, 2014 — C. R. Bard Inc. announced that the U.S. Food and Drug Administration’s (FDA) Circulatory System Devices ...
June 16, 2014 — New appropriate use expert consensus documents developed by the Society for Cardiovascular Angiography ...

June 5, 2014 — Mercator MedSystems is in the midst of its prospective, 300-patient, 30-site DANCE (Dexamethasone to the ...
May 16, 2014 — In a move to expand significantly its portfolio of solutions for peripheral interventions, Boston ...
May 7, 2014 — Women face greater limits on their lifestyle and have more severe symptoms as a result of peripheral ...
April 23, 2014 — Interim results from The Chocolate Bar Registry conducted in the United States demonstrate that use of ...

 August 12, 2014
August 12, 2014