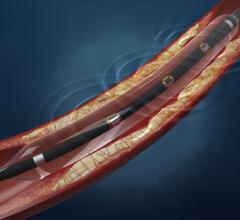
February 6, 2015 — United States hospitals began using a new medical device from Medtronic plc called the In.Pact ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.

January 30, 2015 — ECRI Institute has created a report that offers an overview of drug-eluting balloon (DEB) technology ...
January 16, 2015 — Non-contrast enhanced MR angiography (MRA) techniques have attracted interest in the medical ...
January 13, 2015 — Shockwave Medical announced CE Mark regulatory approval for the company’s Lithoplasty balloon ...
January 9, 2015 — Covidien announced it has received CE Mark approval for its Stellarex drug-coated angioplasty balloon ...
December 31, 2014 — Roxwood Medical Inc. announced U.S. Food and Drug Administration (FDA) clearance for the U.S ...
December 22, 2014 — Advanced Vascular Dynamics (AVD) announced U.S. Food and Drug Administration (FDA) clearance and CE ...
December 17, 2014 — ReFlow Medical Inc. announced U.S. Food and Drug Administration (FDA) clearance for ...
November 20, 2014 — Oxford University researchers presented data that reveal the extent to which smoking causes silent ...
November 17, 2014 — VIVA Physicians, a not-for-profit organization in the field of vascular medicine and intervention ...
Nov. 14, 2014 — Covidien releases 12-month results of the DEFINITIVE AR study, the first randomized study designed to ...
November 11, 2014 — Five-year results from the largest and longest-running clinical trial of a drug-eluting stent for ...

Nov. 11, 2014 — Viva Physicians, a not-for-profit organization in the field of vascular medicine and intervention ...
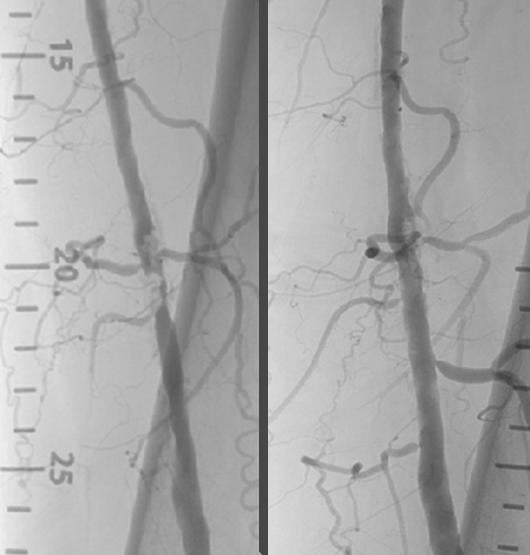
Nov. 11, 2014 — Shockwave Medical announced positive clinical results from Disrupt PAD trial, a single-arm multicenter ...

 February 06, 2015
February 06, 2015