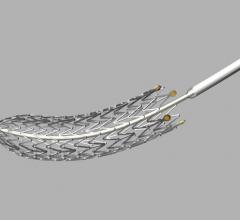
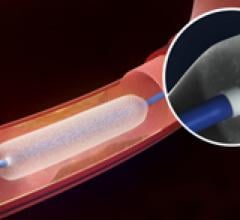
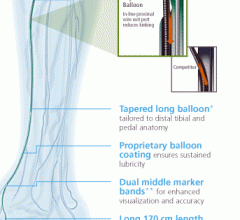
October 14, 2013 — Launching a clinical trial expected to serve as the foundation for global regulatory approvals, a ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
October 11, 2013 – The Boston Scientific OffRoad Re-Entry Catheter System demonstrated excellent performance in ...
October 8, 2013 — Biotronik announced it has completed patient enrollment in the iliac arm of its Bioflex-I trial. Under ...
October 1, 2013 — The U.S. Food and Drug Administration (FDA) approved Medtronic Inc.'s Complete SE (self-expanding) ...
September 3, 2013 — Avinger Inc. has received CE Mark approval for Pantheris – a system that combines directional athere ...

August 28, 2013 — Effective Oct. 1, Cook Medical’s Zilver PTX drug-eluting peripheral stent qualifies for new-technology ...
|
August 28, 2013 — According to Millennium Research Group (MRG), efforts to streamline the regulatory approval process in ... |
August 26, 2013 — Boston Scientific Corp. has completed enrollment in the SuperNOVA trial – a global, single arm ...
August 23, 2013 — At the opening session of the third annual Amputation Prevention Symposium (AMP) in Chicago, Mary L ...
August 19, 2013 — Cook Medical is again shipping its Zilver PTX drug-eluting peripheral stent to medical centers in the ...
July 18, 2013 — Abbott announced that it has entered into an agreement to purchase IDEV Technologies, a privately held ...
June 25, 2013 — Cordis Corp. announced the European CE marking and U.S. Food and Drug Administration (FDA) approval of ...
June 10, 2013 — C. R. Bard Inc. announced the enrollment of the first patient into the Lutonix Below the Knee (BTK) Clin ...
June 4, 2013 — Covidien recently introduced its RapidCross 0.014-inch Rapid Exchange Percutaneous Transluminal ...
May 15, 2013 — The Spectranetics Corporation announced the U.S. Food and Drug Administration's (FDA) approval of its ...


 October 14, 2013
October 14, 2013