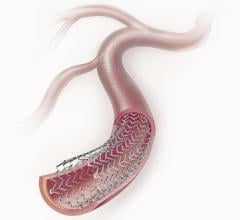
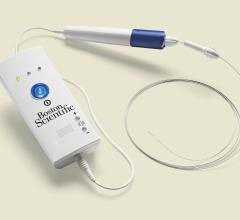
June 21, 2012 — TriReme Medical Inc. announced today it received U.S. Food and Drug Administration (FDA) clearance for ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
June 13, 2012 — Physicians presented at EuroPCR 2012 the results of two multicenter, randomized controlled trials, the ...
June 8, 2012 — Austin Heart, the largest provider of cardiac and vascular services in central Texas, announced its ...
June 5, 2012 — Hansen Medical Inc. received 510(k) market clearance from the U.S. Food and Drug Administration (FDA) for ...
May 23, 2012 - Boston Scientific Corp. announces U.S. Food and Drug Administration approval and market launch of the ...
May 17, 2012 — Boston Scientific Corp. announces CE mark and European market launch of the Innova Self-Expanding Bare ...
May 14, 2012 -- Two types of stent systems are both effective in treating patients who have severe forms of peripheral ...
May 4, 2012 - Angioslide Ltd., a provider of embolic capture angioplasty solutions, announced that it has received U.S ...
May 4, 2012 - A new balloon catheter system could advance the endovascular approach to treating obstructed arteries in ...
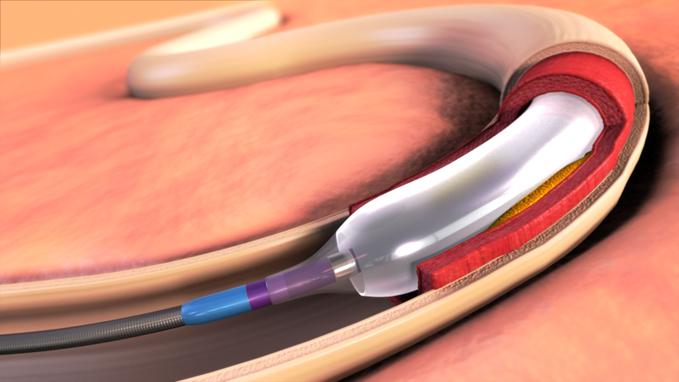
April 17, 2012 –– Expanding its commitment to developing innovative treatments for cardiovascular disease and the ...
March 16, 2012 — The U.S. Food and Drug Administration (FDA) approved the premarket approval (PMA) application for the ...
March 9, 2012 — Avinger Inc. announced the enrollment of the first U.S. patient in the CONNECT II global clinical trial ...
March 6, 2012 — In a development that brings advanced combination therapy treatment of peripheral artery disease (PAD) t ...
March 6, 2012 — Vascular Magnetics announced it has raised $7 million to advance development of a drug delivery system ...
February 9, 2012 — Boston Scientific announced the United States launch of the TruePath CTO device, designed to ...

 June 21, 2012
June 21, 2012