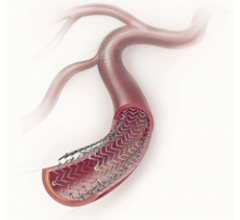
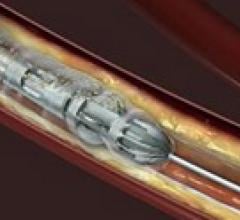
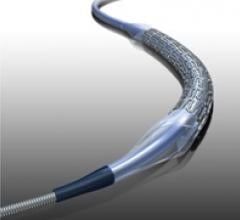
September 15, 2011 — Very positive results were reported in the first account of a clinical study evaluating the ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
September 13, 2011 – An U.S. Food and Drug Administration’s (FDA) will discuss recommendations for Cook Medical Zilver ...
September 8, 2011 – Abbott announced the initiation of ABSORB BTK, an international clinical trial evaluating the safety ...
September 1, 2011 – Medrad Inc. has acquired Pathway Medical Technologies Inc. to strength its vascular interventional ...
August 29, 2011 — The Guidelines on Peripheral Artery Disease (PAD) are the first document produced by the European ...
August 25, 2011 – Two articles in the Society of Interventional Radiology (SIR)'s flagship publication, the Journal of ...
August 25, 2011 – The Munich biotech company apceth started its first Phase I/II clinical study on somatic cell ...
August 10, 2011 – The U.S. Food and Drug Administration (FDA) has approved Abbott’s RX Herculink Elite Renal Stent ...
August 5, 2011 — BridgePoint Medical Inc., a Minnesota-based medical device company, has enrolled its first patient in ...
August 5, 2011 — Vessix Vascular Inc. (formerly known as Minnow Medical Inc.), developer of novel percutaneous ...
August 2, 2011 – The U.S. Food and Drug Administration said it recently cleared Abbott Vascular’s RX Herculink Elite ...
July 20, 2011 — IDEV Technologies Inc. announced the completion of enrollment in the SUPERB trial, a U.S. Food and Drug ...
July 18, 2011 — NSVascular Inc., a newly formed subsidiary of NeuroSigma Inc., has signed an exclusive license with the ...
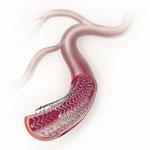
The goal of treating peripheral artery disease (PAD) in the superficial femoral artery (SFA) is limb salvage and ...
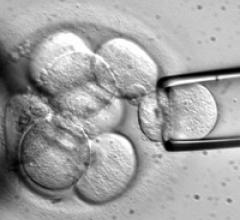
June 29, 2011 – For the first time, human blood vessels grown in a laboratory from donor skin cells have been ...

 September 15, 2011
September 15, 2011