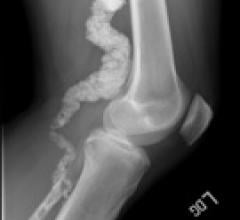
June 20, 2011 — Boston Scientific Corporation today announced the global launch of its Mustang PTA balloon catheter, a ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
June 17, 2011 — The U.S. Food and Drug Administration (FDA) said Boston Scientific has issued a class I recall for its ...
June 3, 2011 - IDEV Technologies announced that data from a long lesion study of 182 patients with significantly ...
As medicine continues its march away from open surgical procedures to minimally invasive and transcatheter treatments ...

May 11, 2011 – In a sign that stem cell therapies for cardiovascular and peripheral artery disease may become mainstream ...
May 10, 2011 – Patients suffering from blockages of the arteries to the kidneys now have access to a new stenting ...
April 13, 2011 – Patient enrollment has begun for the SuperNOVA clinical trial. It will examine the safety and ...
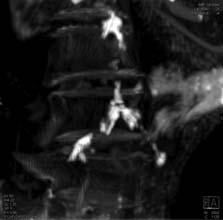
A U.S.-Egyptian research team has uncovered the earliest documented case of coronary atherosclerosis in a ...
April 4, 2011 – The U.S. Food and Drug Administration (FDA) has approved a 210-patient investigation devices ...
March 22, 2011 – The U.S. Food and Drug Administration (FDA) has approved a 210-patient investigation devices ...
March 18, 2011 – Circulating through the bloodstream of every human being is a rare and powerful type of cell, one ...
February 21, 2011 – The U.S. Food and Drug Administration has approved an endoprosthesis device for use on a lower ...
February 16, 2011 – To expand its portfolio of devices for lower extremity peripheral artery disease (PAD) ...
February 4, 2011 – Researchers at the National Institutes of Health's (NIH) Undiagnosed Diseases Program (UDP) ...
January 27, 2010 – The U.S. Food and Drug Administration (FDA) expanded the indication for the Bard Peripheral ...

 June 20, 2011
June 20, 2011