July 18, 2016 — Blood pressure technology company SunTech Medical received U.S. Food and Drug Administration (FDA) ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
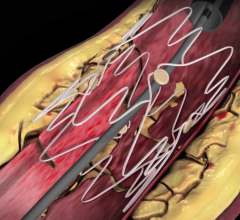
July 13, 2016 — VentureMed Group Ltd., specializing in devices for the endovascular treatment of peripheral arterial ...
June 7, 2016 — Intact Vascular Inc. announced that its Tack Optimized Balloon Angioplasty III (TOBA III) clinical trial ...
June 3, 2016 — Cardionovum GmbH recently announced the completion of enrollment of the RAPID trial. Results will be used ...
May 5, 2016 — Intact Vascular Inc. announced that positive twelve-month results from its Tack Optimized Balloon ...
April 27, 2016 — Veniti Inc. announced the first successful treatment with the Vici Verto Venous Stent System of a ...
April 18, 2016 — The American College of Radiology (ACR), as a member of a coalition of leading medical societies ...
March 29, 2016 — Corindus Vascular Robotics Inc. announced that the U.S. Food and Drug Administration (FDA) has cleared ...
March 28, 2016 — NuCryo Vascular LLC announced the launch of the Next Generation Cryoplasty Inflation device. The device ...
March 22, 2016 — PinnacleHealth CardioVascular Institute enrolled the first patient in Pennsylvania into the TOBA II ...
March 17, 2016 — Boston Scientific announced in late February that the Eluvia Drug-Eluting Vascular Stent System ...
March 16, 2016 — Bioengineers and physicians at the University of California, San Diego have developed a potential new ...
March 3, 2016 — Avinger Inc. announced the company has received 510(k) clearance from the U.S. Food and Drug ...
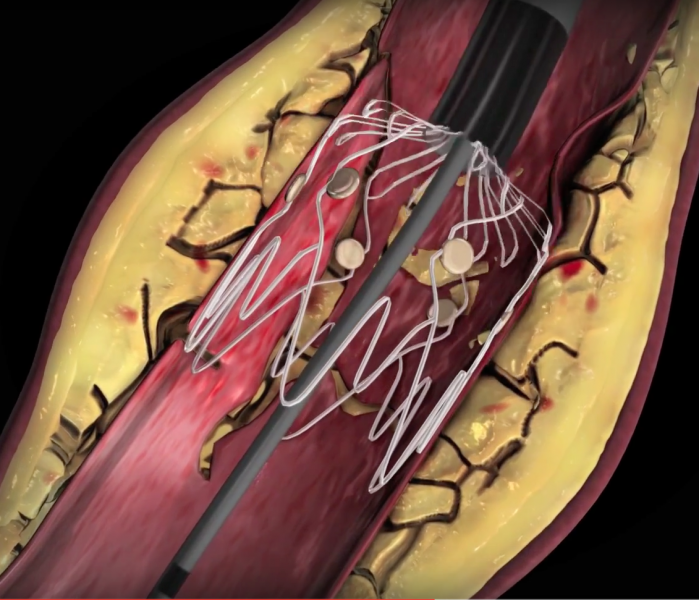
It is estimated that more than 10 million people in the United States are affected by peripheral arterial disease (PAD) ...
February 19, 2016 — Peripheral stents will account for over $4.6 billion in worldwide sales by 2020, according to a new ...

 July 18, 2016
July 18, 2016