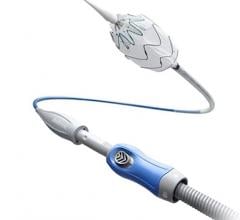
August 13, 2015 — Medtronic plc announced that the first two European patient cases with the Endurant Evo AAA stent graft system were successfully performed at the Erasmus Medical Center in Rotterdam, Netherlands. Both procedures were performed by Prof. Hence Verhagen, M.D., Ph.D., chief of vascular surgery, and his team.
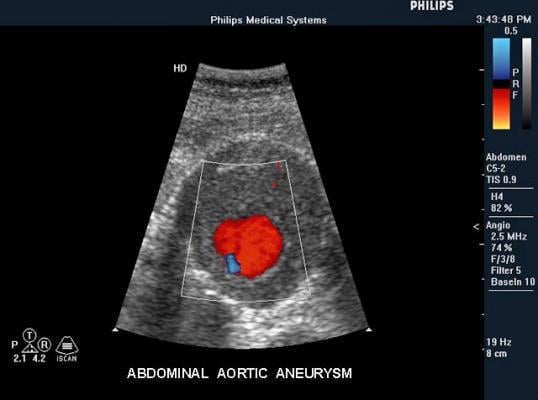
Medtronic began a clinical study in April 2015 to evaluate the safety and effectiveness of the Endurant Evo AAA stent graft system, an investigational medical device designed to expand the applicability of a minimally invasive alternative to open surgical repair for the treatment of abdominal aortic aneurysms.
The first patient was 70 years old and had a 52mm aneurysm. The implant was done with a percutaneous approach thanks to the device’s low profile. The case was successful, with no endoleaks at the final control. The second patient was 74 years old and had a 55mm aneurysm. All the devices were correctly positioned thanks to the 3-piece approach and in-situ sizing capabilities, after a planned coiling and overstenting of one hypogastric artery. The precise deployment resulted in procedural success with no endoleaks at the final imaging.
“The Endurant Evo AAA stent graft system has the potential to improve the treatment for abdominal aortic aneurysms. The system is well designed and the simplified procedure helped to reduce the procedure time,” said Verhagen.
The study will enroll 140 patients with infrarenal abdominal aortic or aortoiliac aneurysms at up to 30 sites in Europe and the United States. All study patients will be treated with the Endurant Evo AAA stent graft system. The study will evaluate safety and efficacy of the new stent graft system. The primary effectiveness objective is to evaluate successful delivery and deployment of the Endurant Evo AAA stent graft system with successful removal of the delivery system during the index procedure. Safety endpoint is defined as the proportion of subjects free from a major adverse event within 30 days post-implantation;
Clinical evidence from the study will be used to seek regulatory approvals required to commercialize the Endurant Evo AAA stent graft system in European, U.S. and other markets around the world.
For more information: www.medtronic.com


 September 18, 2025
September 18, 2025