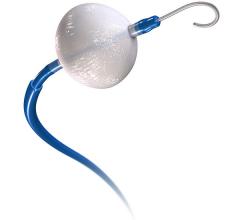
Through a new clinical trial, patients at Harrisburg, Pa.-based PinnacleHealth have access to an investigational device designed to reduce risk of stroke during transcatheter aortic valve replacement (TAVR).
April 11, 2017 — IBA Molecular announced that it has merged with previous acquisition Mallinckrodt Nuclear Medicine LLC ...
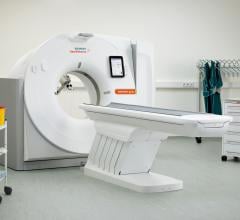
The U.S. Food and Drug Administration (FDA) has cleared the Somatom go. computed tomography (CT) platform from Siemens Healthineers, designed for highly diverse sets of user needs.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
April 10, 2017 — Boston Scientific recently announced a $435 million cash deal to acquire Symetis SA, a privately-held ...
April 10, 2017 — Medtronic recently announced the first enrollments in the STOP Persistent AF clinical trial. The study ...
A discussion with Heart Rhythm Society (HRS) President Michael Gold, M.D., Ph.D., director of cardiology and associate ...
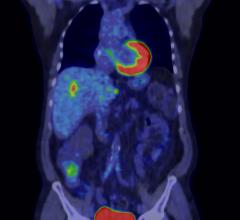
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
David Steenblock, D.O., an osteopathic physician based in San Clemente, Calif., recently discussed his use of hyperbaric oxygen to achieve significant and improved lifestyle outcomes for many stroke patients. The technique involves the therapeutic use of oxygen under pressure to dramatically reduce the effects of stroke and brain injury.
Endologix Inc. announced that the first patients were treated in the Expanding Patient Applicability with Polymer Sealing Ovation Alto Stent Graft (ELEVATE) IDE clinical study.
The Society of Cardiovascular Computed Tomography (SCCT) and Toshiba Medical announced a new partnership dedicated to providing career development opportunities for radiology fellows and residents in the United States. Toshiba Medical’s support will allow 250 U.S.-based fellows and residents to join SCCT at no charge for one year, enhancing their early career opportunities and building their awareness of the growing contributions of cardiovascular computed tomography.
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
PTS Diagnostics announced that the U.S. Food and Drug Administration (FDA) has cleared an expanded top range for measurement of HDL cholesterol (HDL) to 120 mg/dL (3.1 mmol/L) for the company's PTS Panels lipid panel test strips. The PTS Panels lipid panel test strip now has a measurement range of 20 to 120 mg/dL (0.52 to 3.1 mmol/L) for HDL. Additionally, the lipid panel shelf life has increased due to the use of new test strip vials.
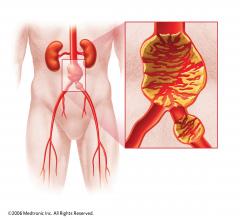
Following repair of abdominal aortic aneurysms (AAA), women appear to have more unfavorable outcomes than men in terms of mortality and morbidity.
April 5, 2017 — GE Healthcare has signed an agreement with HealthTrust, a group purchasing organization headquartered in ...
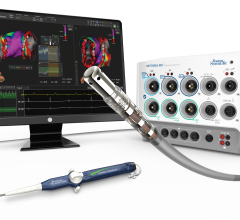
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
This week the Centers for Medicare & Medicaid Services (CMS) convened a panel of the Medicare Evidence Development and Coverage Advisory Committee (MEDCAC) to examine which health outcomes in studies of heart failure treatment technologies should be of interest to CMS. The panel also assessed the growing challenges associated with the changing landscape of evidence generated prior to market authorizations of new and innovative technology. While MEDCAC panels do not make coverage determinations, CMS does benefit from their guidance.
A multi-center study done in the United States and Canada may have global applicability for families with children afflicted with cardiomyopathy. Findings from this large National Institutes of Health (NIH) study, which have important implications in how to treat two types of dilated cardiomyopathy in children, have been published in Circulation Heart Failure, an official journal of the American Heart Association. The paper, representing findings from nearly 100 pediatric cardiology centers, is titled “Differences in Presentation and Outcomes Between Children with Familial Dilated Cardiomyopathy and Children With Idiopathic Dilated Cardiomyopathy: A Report From the Pediatric Cardiomyopathy Registry Study Group.”
Cardiovascular Systems Inc., in partnership with the Cardiovascular Research Foundation (CRF), announced the first subject enrolled in its ECLIPSE clinical trial. The subject was treated by Richard Shlofmitz, M.D., interventional cardiologist and director of the Department of Cardiology at St. Francis Hospital, Roslyn, N.Y. Shlofmitz also treated subjects in CSI’s ORBIT II and COAST clinical studies.

 April 11, 2017
April 11, 2017