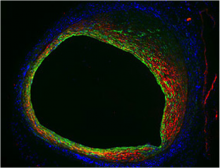
National Institutes of Health researchers have identified a biological pathway that contributes to the high rate of vein graft failure following bypass surgery. Using mouse models of bypass surgery, they showed that excess signaling via the transforming growth factor beta (TGF-Beta) family causes the inner walls of the vein become too thick. This slows down or sometimes even blocks the blood flow that the graft was intended to restore. Inhibition of the TGF-B signaling pathway reduced overgrowth in the grafted veins.
If you enjoy this content, please share it with a colleague
- Read more about Researchers Find Why Many Vein Grafts Fail
- Log in or register to post comments