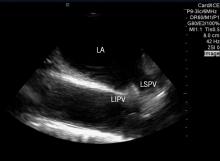
Digging through boxes, waiting for couriers, finding smashed VHS tapes in the mail, jockeying back and forth between multiple modalities. Like at other hospitals across the country, the manual, tape- and film-based workflow in the pediatric cardiology department at Mercy Children’s Hospital in Toledo, Ohio, had stagnated, quadrupling the time it should have taken the cardiologists to read echocardiograms for the hospital and four outreach centers, resulting in a report turnaround time that could be as long as five to seven days.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now