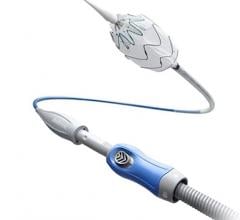
March 4, 2016 — W. L. Gore & Associates (Gore) announced the availability of a new 7.5 cm length of the Gore Viabahn Endoprosthesis with heparin bioactive surface. Gore developed this size in response to physician needs for certain clinical situations. The device is now available in six lengths: 2.5, 5, 7.5, 10, 15 and 25 cm.
In 2013, the U.S. Food and Drug Administration (FDA) granted indication for the Gore Viabahn Endoprosthesis to treat stenosis or thrombotic occlusion at the venous anastomosis of synthetic arteriovenous access grafts. The flexibility of the device allows placement across the elbow and other challenging venous anastomotic locations.
“Selecting the right length device is an important part of getting good outcomes for dialysis access patients. Until now, some cases required choosing between stacking two devices or unnecessarily covering healthy tissue,” said John Ross, M.D., Dialysis Access Institute, Orangeburg Regional Medical Center, Orangeburg, S.C. “The new 7.5 cm length Viabahn improves the ability to land the device with the most accuracy."
For more information: www.goremedical.com


 September 18, 2025
September 18, 2025