Austin Medical Design's Cath Cube is a single-use product for management of guidewires and…
Abiomed Inc. announced it has received 510(k) clearance from the U.S. Food and Drug…
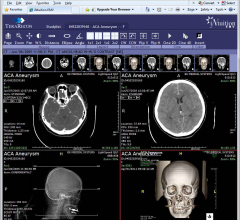
September 10, 2012 — TeraRecon announced the launch of iNtuition Review, iNtuition Enterprise…
September 10, 2012 — In a recent clinical trial, the Solitaire Flow Restoration Device…
September 4, 2012 — Philips Heathcare announced that mVisum Inc.’s STEMI Alert smartphone app is…
August 28, 2012 — Toshiba America Medical Systems Inc. has received U.S. Food and Drug…
August 28, 2012 — NDS Surgical Imaging (NDSsi) is now shipping its new Dome GX4MP…
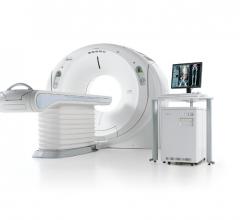
August 23, 2012 — The U.S. Food and Drug Administration (FDA) has cleared Toshiba’s Aquilion RXL…
Philips Healthcare and Corindus Vascular Robotics announced a distribution agreement for…
August 21, 2012 — The U.S. Food and Drug Administration (FDA) granted marketing approval for…
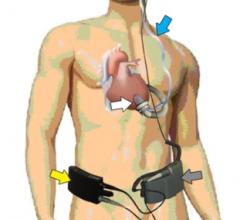
August 17, 2012 — Jarvik Heart Inc., a privately held company that develops and manufactures…
August 17, 2012 — Siemens Healthcare announced an agreement to enhance the syngo…
August 14, 2012 — The U.S. Food and Drug Administration (FDA) has granted 510(k) market…
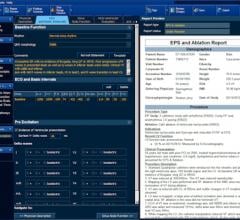
Biosense Webster Inc. launched the new Carto 3 MEM (Multi-Electrode Mapping) Version in the…
McKesson released its Cardiology 13.0 system, which offers a single database solution for…
August 13, 2012 — To improve…
August 9, 2012 — CardioDx Inc. announced that Palmetto GBA, a national contractor that…
Aug. 8, 2012 — The U.S. Food and Drug Administration (FDA) approved the Abbott’s Omnilink Elite …
Aug. 8, 2012 — Vascular Solutions Inc. launched the SuperCross FT, a new flexible-tip version of…
July 31, 2012 — Bracco Diagnostics Inc., the U.S.-based company of the Bracco Group, announced…


 September 13, 2012
September 13, 2012