Feb. 27, 2026 — The U.S. Food and Drug Administration (FDA) has approved Abbott’s CardioMEMS Hero device — a pulmonary ...
Feb. 25, 2026 — Gentuity LLC, a medical technology company specializing in next-generation intravascular imaging devices ...
Feb. 26, 2026 — Eko Health has partnered with Wayne General Hospital in Wayne County, Mississippi, to deploy SENSORA, an ...
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
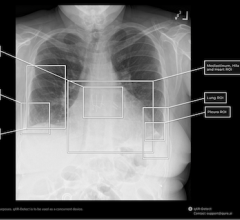
Feb. 26, 2026 — The U.S. Food and Drug Administration (FDA) has given 510(k) class II clearance of qXR-Detect, the ...
Feb. 10, 2026 – AccurKardia, a provider of ECG-based diagnostics technology, recently announced results from a new study ...
Feb. 19, 2026 — Preliminary results from an Italian registry describe the risk profile of women experiencing acute ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Feb. 18, 2026 — Researchers at the Mount Sinai Kravis Children’s Heart Center led a multicenter effort to develop and ...
The American Society of Radiologic Technologists (ASRT) will host a free Virtual Career Fair on March 17, from 4-7 p.m ...
Feb. 14, 2026 — Cardiovascular Research Foundation (CRF) has announced the creation of Complex Coronary Summit, a new ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Feb. 9, 2026 — HTA and MedAxiom have opened applications for the 2026 HeartX program, a cardiovascular-focused ...
Feb. 16, 2026 — MemorialCare Heart & Vascular Institute at Orange Coast Medical Center has earned national recognition ...
Feb. 17, 2026 — The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for Retia Medical's Argos ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Feb. 13, 2026 — Conavi Medical Corp. recently highlighted the publication of new peer-reviewed research titled “Deep ...
Dec. 10, 2025 — The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to HeartBeam, Inc. for its 12 ...
Feb. 11, 2026 —The American Roentgen Ray Society (ARRS) has announced the following radiologists, as well as their ...


 February 27, 2026
February 27, 2026