April 4, 2022 – Impulse Dynamics, a global medical device company dedicated to improving the lives of people with heart ...
April 3, 2022 — egnite, Inc., an advanced technology digital health company focused on providing artificial intelligence ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
April 3, 2022 — A new drug shows promise in providing relief to cardiomyopathy patients experiencing shortness of breath ...
April 3, 2022 — Silence Therapeutics, a leader in the discovery, development and delivery of novel short interfering ...
April 2, 2022 — Royal Philips, a global leader in health technology, today announced the launch of Ultrasound Workspace ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
April 2, 2022 — Aidoc, a leading provider of healthcare AI solutions, announced that it received FDA 510(k) clearance ...
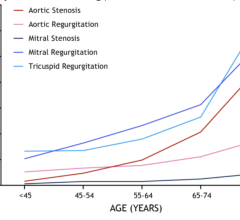
April 1, 2022 — A new joint guideline published today from the American College of Cardiology, the American Heart ...
April 1, 2022 — Cardiologs announced a poster presentation at the 2022 American College of Cardiology (ACC) meeting in ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
April 1, 2022 — Royal Philips, a global leader in health technology, announced new additions to its innovative ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
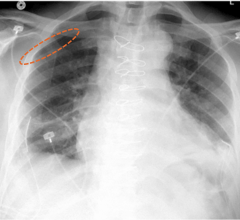
March 31, 2022 — In accordance with its commitment to patient safety, Medtronic recently voluntarily recalled a subset ...
Here is what you and your colleagues found to be most interesting in the field of cardiology during the month of March ...


 April 04, 2022
April 04, 2022



![Royal Philips, a global leader in health technology, today announced the launch of Ultrasound Workspace [OLK1] at the American College of Cardiology’s Annual Scientific Session & Expo (ACC 2022).](/sites/default/files/styles/content_feed_medium/public/philips-ultrasound-workspace-cardiology-diagnosis.jpeg?itok=VFUpaaT2)