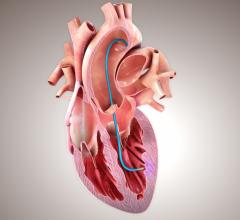
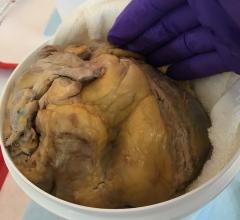
Specially processed, non-crosslinked, acellular extracellular matrix (ECM) implanted on a heart.
Over the last decade, cardiac stem cell therapy has shown tantalizing but as yet unrealized potential as a treatment for the left-ventricular dysfunction (LVD) that develops subsequent to myocardial infarction (MI). Experimentally, somatic stem cells of various lineages, as well as pluripotent stem cells, have been induced to transform into contractile myocytes expressing cardiac-specific proteins. The heart is also known to contain its own supply of multipotent stem cells. Despite the ready availability of these raw materials, there has been little progress in efforts to identify a cell-therapy approach that can redirect maladaptive cardiac remodeling toward constructive pathways, support production of de novo myocardial tissue, and ultimately prevent or reverse post-MI LVD and congestive heart failure (CHF) [1].
Cellular Micro-Environment Determines Constructive Remodeling Pathways
It is becoming increasingly apparent that isolated stem cells, whether intrinsically or extrinsically derived, cannot respond in a constructive fashion to the conditions encountered in inflamed, diseased or healing tissues. Growing evidence suggests that the pathological micro-environment within the infarct – specifically, the destruction of the native myocardial extracellular matrix – exerts a critical negative influence on the myocardium’s regenerative capacity. Immediately following MI, in situ, myocardium-specific niche stem cells become activated. Simultaneously, marrow-derived progenitor cells begin homing to the site of injury. These potentially regenerative cells accumulate in the ischemic periphery of the infarct, but in the absence of a structurally and biochemically functional extracellular matrix, they proliferate ineffectively into fibroblasts and myofibroblasts. The resulting formation of nonfunctional scar tissue has not been overcome by external stimulation with proliferative factors or supplementation with ex vivo expanded, re-infused stem cells.
Once thought of as inert scaffolding, the extracellular matrix is now known to be a complex, three-dimensional, inductive informational system essential to normal tissue physiology. Together with its resident cells, the extracellular matrix participates in a two-way exchange of information that has been termed “dynamic reciprocity” [2]. It provides stem cells with a recruitment signal, an attachment point and environmental context within which to proliferate, metabolize and mature phenotypically into functional myocytes and surrounding structural tissues. In response, the maturing cells lay down new, tissue-specific extracellular matrix, which recruits and instructs additional stem cells in an iterative process that, in damaged tissue, can enable constructive remodeling toward a normally functioning tissue.
External Use of Extracellular Matrix to Restore the Cellular Microenvironment
A number of injectable biomaterials, including fibrin glue, collagen, self-assembling nanopeptides and alginate, have been investigated for their ability to serve as scaffolding for cellular regeneration. While these have shown some promising improvements in healing and remodeling, none are composed of or contain an extracellular matrix with the capacity to provide architecture capable of supporting the development of site-specific tissue.
A promising new route of investigation is based on the use of a specially processed, non-crosslinked, acellular extracellular matrix (ECM) product derived from porcine small-intestinal submucosa (SIS) [3, 4]. This biomaterial is commercially available in sheet form for repair of human cardiac tissues and as a patch to facilitate reconstruction of the pericardium following open-heart surgery (CorMatrix ECM for cardiac tissue repair and for pericardial closure).
Sturdy enough for intracardiac implantation but completely bioresorbable, the ECM sheet product has been shown to recruit marrow-derived stem cells and increase the density of new cardiomyocytes at the site of ECM implantation [3-5]. It provides a unique environment in which stem cells can attach, migrate and mature phenotypically. Furthermore, it is less prone to induce an inflammatory response, less subject to calcification and probably more resistant to infection than synthetic patch materials, as it integrates synergistically with, and eventually resorbs into, the underlying tissue. This material has been implanted during nearly 30,000 human cardiac repair procedures, including suture line reinforcing, buttressing for soft tissue re-approximation, pledgets, cannulation sites, structural reinforcement, septal defect repairs, and pericardial closure. It is priced competitively compared with synthetic and biosynthetic surgical patch material and eligible for reimbursement.
With respect to the development of new therapies for MI and CHF, recent preclinical studies have shown that an injectable form of the ECM product recruits c-Kit+ (marrow-derived) stem cells, myofibroblasts and macrophages after MI and induces the phenotypic differentiation of stem cells into cardiomyocytes. In a rat model of MI, these cellular infiltration and differentiation phenomena correlate directly with improvements in left ventricular function [5].
Additional preclinical work is ongoing, with in-human trials to test the safety and therapeutic potential of the injectable ECM to be planned based on the results.
Should the results of human trials support the findings of the preclinical studies, ECM-based products may logically become an important inclusion in stem cell-based therapies for MI with LVD, and for the correction of CHF. The hope is that treatment with ECM-based products, alone or in conjunction with cellular transplantation, would finally harness the regenerative power of stem cells for true, permanent disease modification, rather than the temporary palliation of symptoms offered by current pharmacologic, device-based and surgical treatment options.
Future Directions for ECM Therapy
Other investigational applications for the ECM product include a potential therapeutic effect in postoperative atrial fibrillation (AF). A recent retrospective, multicenter, case-control study found that use of the ECM (sheet form) to enable pericardial closure following primary, isolated coronary artery bypass graft (CABG) resulted in a 54 percent reduction in the relative risk of developing new-onset postoperative AF (20 percent absolute risk reduction) as compared to patients with their pericardium left open [6]. Hypothetical explanations for this finding include physical sequestration of the heart from shed mediastinal blood and its pro-inflammatory components; limitation of postoperative volume shifts, atrial stretch and secondary structural alterations; and/or intrinsic biological properties of the ECM itself. A prospective, multicenter, randomized controlled trial is being conducted to further investigate these hypotheses.
Pre-clinical studies evaluating a number of additional interventional and surgical delivery platforms are also under way.
Conclusion
MI with LVD and CHF have thus far proven intractable to conventional models of stem cell therapy. A greater understanding of the normal myocardial microenvironment, and development of biomaterials that restore this environment in the infarcted heart, may soon enable the full realization of stem cell therapy’s regenerative potential.
Editor’s note: Thomas F. Deering, M.D., FACC, FACP, FHRS, is chief of the Piedmont Heart Institute Arrhythmia Center, Atlanta, Georgia
Related Content About Stem Cell Therapies to Restore Heart Function
FDA Says Adult Stem Cell Research Shows Promise
Stem Cell Therapy Holds Promise for Treating Most Severe Cases of Angina
3-D Printed Patch Can Help Mend "Broken" Heart
Cardiac Regeneration After MI: Role of the Extracellular Matrix
Resurrecting Dead Heart Tissue
References:
1. Mummery CL, Davis RP, Krieger JE. “Challenges in using stem cells for cardiac repair.” Sci Transl Med, 2010; 2(27): 27ps17.
4. Badylak S, Obermiller J, Geddes L, Matheny R. “Extracellular matrix for myocardial repair.” Heart Surg Forum, 2003; 6(2): E20–6.
6. Boyd WD, Johnson WEr, Sultan PK, Deering TF, Matheny RG. “Pericardial reconstruction using an extracellular matrix implant correlates with reduced risk of postoperative atrial fibrillation in coronary artery bypass surgery patients.” Heart Surg Forum, 2010; 13(5): E311–6.


 November 19, 2021
November 19, 2021