May 21, 2015 – A preliminary study of a new electrophysiology (EP) dipole density mapping system using a specialized intracardiac ultrasound basket catheter for electro-mapping, was showen to offer four times higher resolution than standard voltage mapping. Researchers also said the new mapping modality represents the electromapping data on computed tomography (CT) quality anatomy. The data suggest it may open the possibility to map irregular arrhythmias with more precision. The data was presented at Heart Rhythm Society’s 2015 meeting.
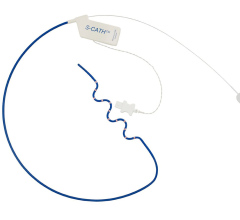
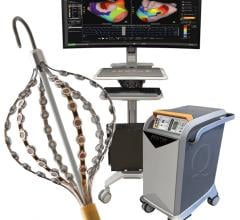
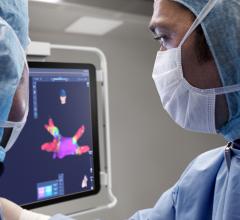
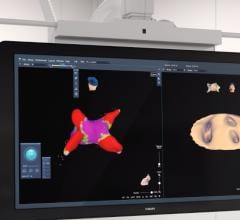
Acutus Medical presented data that shows its AcQMap high resolution, imaging and mapping system generates a complete heart chamber electrical map that provides a four-fold improvement in resolution to record and display heart tissue function as compared to the standard voltage-based mapping used in patients with atrial flutter. Today’s EP mapping systems used during catheter ablation procedures capture and display a voltage field generated by heart tissue during activation. This voltage field records over a large area of heart tissue at each catheter placement and what is seen by the EP is an average of all the tissue around the catheter, reducing the resolution. This makes it difficult for the EP to see the heart tissue in its “true” resolution and limits the possibility to map accurately and simply diagnosis irregular and complex arrhythmias.
“The higher resolution image generated by the AcQMap System may open the possibility to truly map irregular heart rhythms with more precision and eliminate the unnecessary guesswork and lengthy procedures common today among even the most experienced electrophysiologist,” said Stephan Willems, M.D., Ph.D., University Heart Center, University Hospital Eppendorf, Hamburg, Germany. “The data also shows the potential of the AcQMap System to change clinical practice and improve treatment outcomes in patients with irregular and complex heart rhythm disorders.”
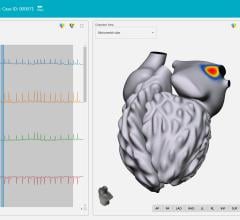
The AcQMap system is designed to generate real-time, 3-D images of the heart chamber with quality equivalent to a CT or MRI in a matter of minutes. The system can detect and display both the standard voltage based maps as well as the higher resolution dipole density maps. The result of combining highly accurate imaging with higher resolution mapping capabilities enables EP’s to identify and locate the responsible tissue for the irregular heart rhythm, target treatment with pinpoint accuracy and minimize heart ablation.
Study Data
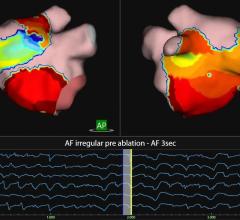
Data were collected from five of eight patients enrolled with atrial flutter to determine the difference in resolution of a dipole density map generated by the AcQMap System, to that of a standard voltage-based map.
The AcQMap system was used to measure cardiac voltage, apply its dipole density algorithm and display electrical activation on the ultrasound constructed 3-D anatomy, in real-time. A dipole density map and voltage-based map were created for each patient to show sinus rhythm, septal and lateral pacing and atrial flutter. The depolarized surface areas of each map were compared by study investigators and results showed that during atrial flutter, mean depolarized area for dipole density maps was 26.6 cm2, significantly less than the voltage based maps, which were 83.2 cm2. Overall the voltage-based map to dipole density map ratio was 4.1 for atrial flutter; 4.7 for SR; and 4.5 for pacing.
For more information: www.acutusmedical.com





 September 05, 2025
September 05, 2025