Dec. 8, 2025 — In November, The Lancet published an international expert consensus review, “Contemporary, non-invasive ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
Nov. 25, 2025 — Remington Medical has received U.S. Food and Drug Administration (FDA) 510(k) clearance and announced ...
Dec. 3, 2025 — Atraverse Medical, a medical device company developing next-generation left-heart access technology, has ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
Dec. 3, 2025 — Recross Cardio Inc., a structural heart company developing next-generation membrane sealing technology ...
Dec. 2, 2025 — At RSNA 2025, Siemens Healthineers announced Syngo.CT Coronary Cockpit1, a new software solution within ...
Nov. 24, 2025 — InspireMD, Inc., developer of the CGuard Prime carotid stent system for the prevention of stroke ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
Nov. 11, 2025 -— Integra LifeSciences Holdings Corp. has announced the FDA 510(k) clearance for use of its CUSA Clarity ...
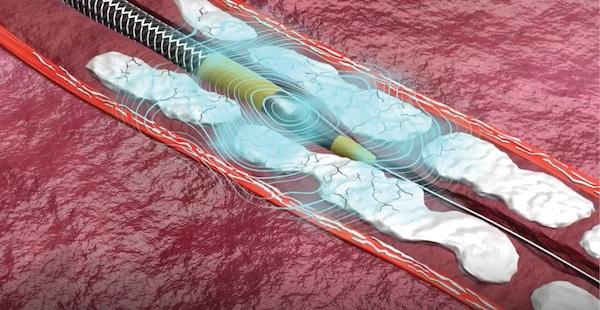
Since receiving FDA approval in 2016, intravascular lithotripsy (IVL) systems have grown in popularity among ...
Nov. 11, 2025 — FastWave Medical has successfully completed enrollment in its 30-patient coronary feasibility study and ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
Nov. 10, 2025 — Sentante, a med-tech robotics company, has successfully demonstrated a first-of-a-kind remote stroke ...
Nov. 4, 2025 — Amplitude Vascular Systems (AVS), a medical device company focused on treating calcified arterial disease ...
Nov. 3, 2025 — Penumbra, Inc. has announced additional results of the STORM-PE randomized controlled trial (RCT), which ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
Nov. 4, 2025 – Johnson & Johnson MedTech has announced the one-year results in patients treated with its Shockwave ...
Oct. 28, 2025 — Results from the first-of-its-kind randomized PROCTOR trial found that a strategy of saphenous vein ...
Oct. 30, 2025 — Recor Medical and its parent company, Otsuka Medical Devices presented the results from two clinical ...

 December 11, 2025
December 11, 2025