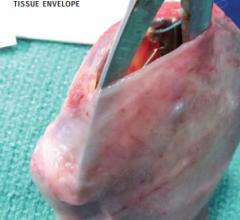
There have been several recent advancements in implantable cardioverter defibrillator (ICD) technology to extend battery ...
Implantable Cardioverter Defibrillator (ICD)
This channel includes news and new technology innovations for implantable cardioverter defibrillators (ICD) used to treat tachycardia arrhythmias and heart failure. This includes cardiac resynchronization therapy defibrillators (CRT-D).

February 5, 2015 — Boston Scientific announced the launch and the first U.S. implant of its line of Extended Longevity ...
January 30, 2015 — Medtronic announced new results from the PainFree SST and Shock-Less clinical studies published in ...
January 7, 2015 — Biotronik announced the publication of new findings from the REPLACE Registry in Circulation ...
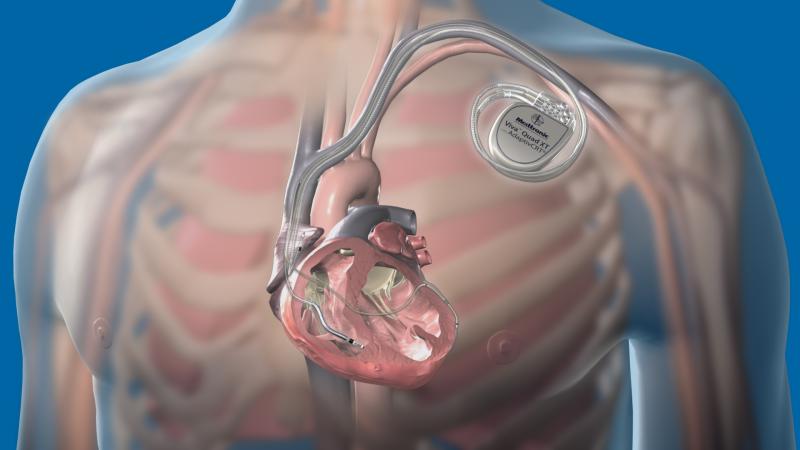
December 17, 2014 – Medtronic Inc. announced the U.S. Food and Drug Administration (FDA) approval and commercial launch ...
December 11, 2014 — A new study evaluating Optim-insulated implantable cardioverter defibrillator (ICD) leads found low ...
December 5, 2014 — Most patients with implantable cardioverter defibrillators (ICDs) haven’t thought about device ...
November 14, 2014 — Boston Scientific Corp. announced that its subcutaneous implantable defibrillator (S-ICD System) ...
September 12, 2014 — Ecolab Healthcare announced the availability of the PD220 patient drape, the first to market ...

September 9, 2014 — Use of a quadripolar left ventricular (LV) lead instead of a bipolar option during cardiac ...
August 27, 2014 — Fysicon received approval from the U.S. Food and Drug Administration (FDA) to market Fysicon DataLinQ ...
August 11, 2014 — Medtronic announced it has received U.S. Food and Drug Administration (FDA) approval for the Attain ...
July 10, 2014 — Biotronik announced that the first device patients to undergo full-body magnetic resonance imaging (MRI) ...
May 28, 2014 — Biotronik announced that the U.S. Food and Drug Administration (FDA) has approved a significant expansion ...


 February 23, 2015
February 23, 2015