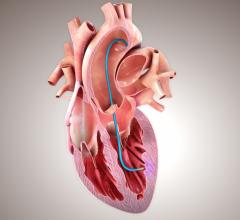
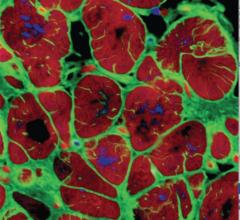
July 16, 2014 — CorMatrix Cardiovascular and the Central Clinical Hospital of the Ministry of Interior in Warsaw (Centralny Szpital Kliniczny MSW) of Warsaw, Poland, announced the successful enrollment and treatment of five patients in the first clinical study evaluating the CorMatrix ECM delivered trans-epicardially for the improvement of myocardial function in patients at risk or suffering from congestive heart failure (CHF).
The RESTORE study is being conducted at the Central Clinical Hospital of the Ministry of Interior in Warsaw, Poland under the direction of study investigator Dr. Piotr Suwalski, chief of the Department of Cardiac Surgery. The objective of this study is to evaluate the safety of the CorMatrix ECM delivered trans-epicardially with a proprietary delivery system to patients with left ventricular ejection fraction (LVEF) 25 to 40 percent during coronary artery bypass grafting (CABG). The endpoints of the study include device related safety and improvement in global ventricular function. Echocardiograph and magnetic resonance imaging (MRI) data will be evaluated by Yale Cardiovascular Research under the direction of Alexandra Lansky, M.D. Patients will be followed for 18 months, with interim assessments being conducted at six months and 12 months post treatment.
Heart failure (HF) or congestive heart failure (CHF) represents one of the largest unsolved medical problems today, with more than 5 million patients in the United States alone, and this number is expected to double over the next 10 years. In 2012, the direct and indirect cost of HF in the United States was estimated to be $31 billion. Globally, the incidence of HF is increasing with more than 1 million new cases diagnosed annually. Current treatment options for HF are limited to pharmacological therapy, cardiac resynchronization therapy, mechanical circulatory support devices and heart transplant. In advanced HF, transplant offers the best opportunity for long-term survival, but is restricted to select patients and the number of available donor organs is limited and cannot meet the growing demand. These circumstances have promoted mechanical circulatory support devices as a treatment option aimed at restoring cardiac output. Left ventricular assist devices (LVAD), which mechanically unload blood volume from the failing left ventricle into the aorta, have become a standard therapeutic option for bridging to transplantation or for permanent use in patients not eligible for heart transplantation with end-stage HF. Recently the bridge-to-transplantation (BTT) trial/ADVANCE and ADVANCE CAP by Slaughter and Aaronson and another publication from Starling reveal BTT outcomes are equivalent to heart transplant at one and two years. Additionally, a paper by Kirklin shows that destination therapy (DT) outcomes at two years are also equal to transplant).
CorMatrix has developed an extracellular matrix device and an intra-myocardial delivery device that enables efficient implantation of the CorMatrix ECM directly into damaged cardiac tissue. Intra-myocardial delivery of the CorMatrix to mechanically stabilize, repair and recover infarcted regions of the heart could provide myocardial recovery for these heart failure patients. The CorMatrix ECM is an implantable material derived from porcine small intestine submucosa (SIS) that has been shown to provide a biologically and mechanically favorable scaffold for cell incorporation, differentiation, and proliferation. The proprietary delivery system enables targeted implantation and uniform distribution of the CorMatrix ECM to the ischemic regions of the myocardium to maximize recovery.
Suwalski commented that "it is very exciting to take part in a study like the RESTORE Study that has the potential to provide data in support of what would be the first time the potential of a patient’s own stem cells were harnessed for the treatment or prevention of congestive heart failure by restoring a natural matrix with the regenerative power of the heart. The CorMatrix ECM treatments and the delivery system provided safe and easy application of the CorMatrix ECM to the patients."
Mark Slaughter, director of the Heart Transplant and Mechanical Assist Device program at Jewish Hospital & St. Mary’s HealthCare, in Louisville, Ky., as well as chair of the Department of Cardiovascular and Thoracic Surgery at the University of Louisville added, "this First in Human study follows pre-clinical work that we’ve conducted at the University of Louisville demonstrating that CorMatrix ECM may provide a favorable environment to enhance myocardial regeneration and promote sustained myocardial recovery. The RESTORE Study will further clarify this question and set the bar for future CHF technologies.”
For more information: www.cormatrix.com


 November 19, 2021
November 19, 2021