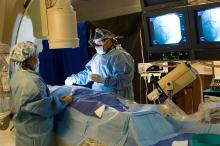
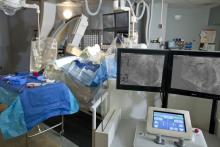
August 15, 2016 – Edwards Lifesciences announced the U.S. Food and Drug Administration (FDA) approved of the advanced Edwards Intuity Elite valve system, a rapid deployment device for surgical aortic valve replacement. Built on the Perimount tissue valve platform and incorporating innovations from transcatheter heart valves, the the new valve is designed to facilitate minimally invasive surgery and streamline complex aortic valve replacements, thereby offering a cutting-edge treatment option for patients with aortic valve disease. The device is now commercially available.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now