Oct. 22, 2025 -- Medical robotics innovator Noah Medical presented two new data sets highlighting the clinical and ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
Oct. 29, 2025 — FastWave Medical presented new first-in-human (FIH) and pre-clinical data for its Sola coronary laser ...
Oct. 27, 2025 — At the annual Transcatheter Cardiovascular Therapeutics (TCT 2025) meeting in San Francisco, Royal ...
Venous thromboembolism (VTE), and pulmonary embolism (PE) in particular, are among the most complex and dangerous acute ...
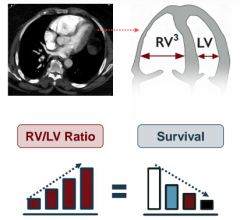
Oct. 27, 2025 — Results from the PREVUE-VALVE study suggest that there are currently at least 4.7 million people aged 65 ...
Oct. 28, 2025 — People who underwent a minimally invasive procedure to have their heart’s aortic valve replaced had ...
Oct., 2025 — Elucid, an AI medical technology company focused on providing physicians with a more precise view of ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
Oct. 25, 2025 — Medtronic plc has announced the launch of the Stedi Extra Support guidewire, designed to enhance ...
Oct. 27, 2025 — Elixir Medical, a developer of technologies to treat cardiovascular disease, has announced new clinical ...
Oct. 23, 2025— Emboline, Inc., a provider of full-body embolic protection devices for transcatheter procedures, thas ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
Oct. 26, 2025 – Jupiter Endovascular, Inc., a medical technology company developing a new class of endovascular ...
Oct. 27, 2025 – Penumbra, Inc. has announced the results of the STORM-PE randomized controlled trial (RCT), which found ...
Oct. 21, 2025 — SpectraWAVE, Inc., a medical imaging company focused on improving the treatment and outcomes for ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
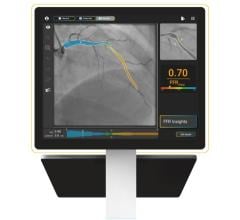
Oct. 22, 2025 — Heartflow, Inc. has introduced Heartflow PCI Navigator, the newest addition to the Heartflow One ...
Oct. 22, 2025 — Qure has announced its latest (510) K clearance from the US Food and Drug Administration (FDA).
The ...
Oct. 22, 2025 — Nyra Medical, a leading innovator in structural heart therapies, has announced the initiation of its ...


 November 03, 2025
November 03, 2025