June 18, 2024 — Elixir Medical has announced the company’s novel bioadaptive implant, DynamX Sirolimus-Eluting Coronary ...
FDA
June 17, 2024 — Elutia Inc., a pioneer in drug-eluting biomatrix products, today announced that its Antibiotic-Eluting ...
June 13, 2024 — Xeltis, a leading developer of transformative implants that enable the natural creation of living and ...
June 13, 2024 — The U.S. Food and Drug Administration (FDA) announced that Teleflex, and their subsidiary Arrow ...
June 12, 2024 — Royal Philips, a global leader in health technology, announced the first implant of the Duo Venous Stent ...
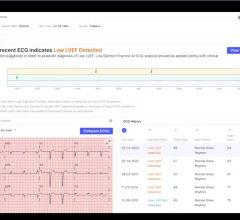
May 21, 2024 — Anumana, a Cambridge, MA-based artificial intelligence (AI)-driven health technology company and ...
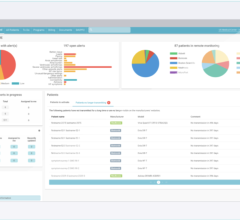
May 17, 2024 — Implicity, a leader in remote patient monitoring and cardiac data management solutions, announced it has ...
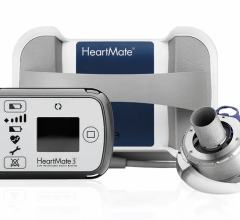
May 15, 2024 — The U.S. Food and Drug Administration (FDA) announced that Abbott is recalling the HeartMate 3 LVAS by ...
May 8, 2024 — The US Food and Drug Administration (FDA) is alerting health care providers and facilities about our ...
May 8, 2024 — 4C Medical Technologies, Inc. ("4C Medical"), a medical device company dedicated to advancing minimally ...

Clinical trials and innovative technology took center stage during the month of April, racking up some record number ...
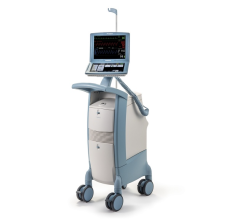
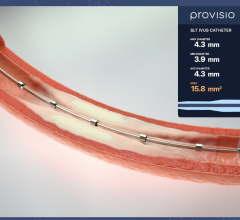
April 25, 2024 — Provisio Medical announced FDA 510(k) clearance of the Provisio SLT IVUS System. Sonic Lumen Tomography ...
April 25, 2024 — Atlantic Health System’s Morristown Medical Center treated the first patient in New Jersey using Edward ...

 June 18, 2024
June 18, 2024