June 04, 2025 — HeartSciences Inc. has announced that the U.S. Food and Drug Administration has granted Breakthrough ...
FDA
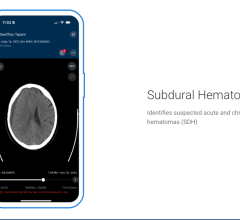
June 12, 2025 — Viz.ai recently announced it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for ...
June 5, 2025 – Penumbra, Inc. has announced the U.S. Food and Drug Administration (FDA) clearance and launch of the Ruby ...
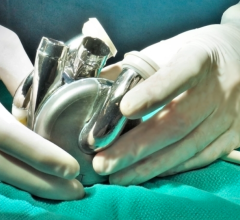
May 30, 2025 — BiVACOR, a clinical-stage medical device company developing the world’s first titanium Total Artificial ...
May 27, 2025 — Abbott has announced the U.S. Food and Drug Administration (FDA) has approved the company's Tendyne ...
May 19, 2025 — Gradient Denervation Technologies recently announced the company’s pulmonary denervation system has ...
May 13, 2025-- GE HealthCare recently announced the U.S. Food and Drug Administration (FDA) has approved a pediatric ...
May 19, 2025 - Arineta, a provider of cardiovascular imaging solutions, recently announced that its SpotLight Duo ...
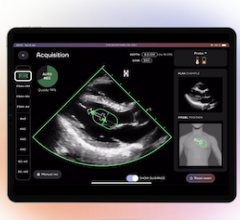
May 14, 2025 — BrightHeart, a provider of artificial intelligence (AI) solutions for pediatric cardiology and obstetrics ...
April 22, 2025 — Orchestra BioMed Holdings, a biomedical company accelerating high-impact technologies to patients ...
April 25, 2025 – Medtronic plc, a global leader in healthcare technology, recently received U.S. Food and Drug ...
April 21, 2025 — Stereotaxis, a provider of surgical robotics for minimally invasive endovascular intervention, will ...
April 15, 2025 – DESKi recently announced FDA clearance for HeartFocus, its AI-enabled heart exam software that empowers ...
April 9, 2025 — Idorsia Ltd recently announced that the US Food & Drug Administration (FDA), after having released ...
April 7, 2025 — CardioVia, a medical device company specializing in advanced cardiac care solutions, has received U.S ...

 June 12, 2025
June 12, 2025