Janssen Pharmaceuticals Inc. announced the U.S. Food and Drug Administration (FDA) has approved…
November 6, 2012 — Vascular Solutions launched the R-Band radial…
November 6, 2012 — Toshiba America Medical Systems Inc. has expanded its Assurance Program to…
The Philips Healthcare Ambient Experience integrates architecture, design and enabling…
November 1, 2012 — TomTec Imaging Systems GmbH announced U.S. Food and Drug Administration (FDA…
November 1, 2012 — Terumo Interventional Systems announced the nationwide availability of its…
Maquet Cardiovascular received 510(k) clearance from the U.S. Food and Drug Administration (FDA…
The Philips IntelliSpace Portal is an advanced visualization solution designed to simplify the…
October 29, 2012 — At RSNA 2012, GE Healthcare will unveil innovations in…
October 29, 2012 — Brainlab released Buzz Digital OR, a major step forward in information…
Covidien announced final 12-month results from its DEFINITIVE LE (Determination of Effectiveness…
October 25, 2012 — InterSystems Corp., a provider of software for healthcare interoperability,…
NEC Display Solutions of America announced the U.S. Food and Drug Administration (FDA) 510(k)…
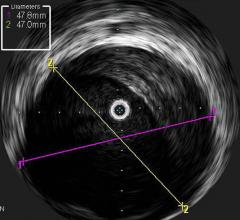
October 19, 2012 — Volcano Corp. announced it has received both U.S. Food and Drug…
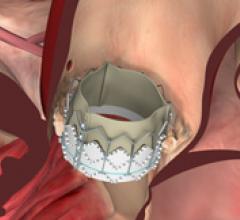
Edwards Lifesciences announced today it received approval from the United States Food and Drug…
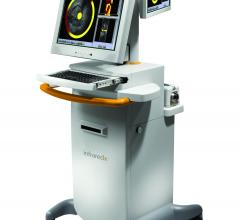
Infraredx Inc., a medical device company, and Royal Philips Electronics announced they have…
October 18, 2012 — Claron Technology has introduced Withinsight Framework (WIF) version 2.1,…
October 15, 2012 — Cardiac Science has unveiled its Burdick 4250 Holter recorder — a wearable…
Surefire Medical Inc. has received U.S. Food and Drug Administration (FDA) 510(k) clearance to…
October 12, 2012 — Estech, a provider of minimally invasive cardiac ablation devices, last month…


 November 07, 2012
November 07, 2012