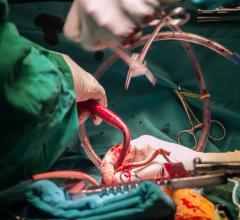
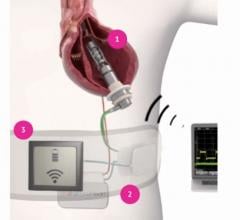
January 13, 2021 — FineHeart, a preclinical-stage medical device company developing a novel left ventricular assist ...
Cardiovascular Surgery
This channel includes news and new technology innovations for cardiac surgery. This includes coronary artery bypass grafts (CABG) and surgical valve replacements.
January 6, 2020 — A new Weill Cornell Medicine found the risk of dying seven years after coronary artery bypass graft ...
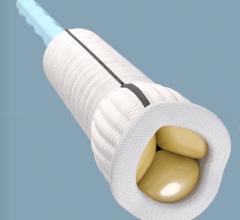
December 14, 2020 - Foldax Inc. today announced that the U.S. Food and Drug Administration (FDA) has granted an ...
December 2, 2020 — The Centers for Medicare and Medicaid Services (CMS) finalized updates to Medicare coverage policies ...
November 18, 2020 — The primary results from the RIVER Trial, Rivaroxaban for Valvular Heart disease and Atrial ...
November 16, 2020 — Atrial fibrillation (AF), an irregular heartbeat that can increase the likelihood of stroke, was ...
October 2, 2020 — The new cardiac hybrid operating room (OR) at London Health Sciences Centre (LHSC) in London, Ontario ...

August 21, 2020 — The U.S. Centers for Medicare and Medicaid Services (CMS) proposed updates to coverage policies for to ...
August 10, 2020 - The Guerin Family Congenital Heart Program in the Smidt Heart Institute at Cedars-Sinai is expanding ...
July 15, 2020 — The U.S. Food and Drug Administration (FDA) granted clearance for the Edwards Lifesciences Corp. Konect ...
June 23, 2020 – Heart valve start-up Foldax is looking to reinventing several aspects of the prosthetic heart valve ...
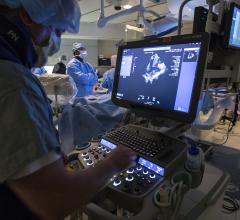
June 4, 2020 — Intra-operative transesophageal echocardiography (TEE) is a versatile diagnostic and monitoring tool used ...

The American College of Cardiology (ACC) together with other North American cardiovascular societies issued a framework ...
May 27, 2020 — Carmat, a developer of the of a next generation advanced total artificial heart, announces the first ...
May 1, 2020 — Abiomed, maker of the Impella heart pump, has acquired Breethe, developer of a novel extracorporeal ...

 January 13, 2021
January 13, 2021