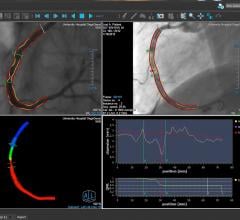
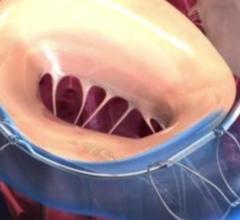
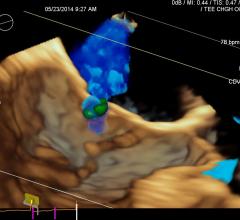
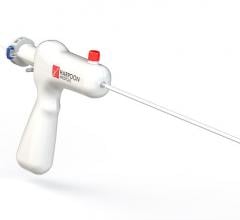
A discussion with William W. O’Neill, M.D., medical director, Center for Structural Heart Disease, Henry Ford Hospital ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
November 8, 2017 — Physicians at UnityPoint Health Methodist, Peoria, Ill., are now offering fast, safe and accurate int ...
John Rhodes, M.D., co-director of the adult congenital heart program, Medical University of South Carolina, is the ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
Juan Granada, M.D., Cardiovascular Research Foundation president and chief executive officer, shares his insights on ...

November 8, 2017 – New results from the HARMONEE Japan/U.S. Registration Trial, reported by in a first report ...
November 8, 2017 — Investigators in the FAVOR II China and FAVOR II Europe-Japan studies recently presented their ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
November 8, 2017 — Medtronic plc recently presented new clinical research to support the positive clinical performance ...
Philippe Genereux, M.D., co-director of the structural heart program at the Gagnon Cardiovascular Institute at ...
Stephen Worthley, MB, BS, Ph.D., director of cardiac catheterization, University of Adelaide, Australia, presents the ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
A discussion with Ajay Kirtane, M.D., SM, director of the cardiac catheterization laboratories at New York-Presbyterian ...

November 7, 2017 – A new study has found that a pioneering device to repair heart valves is safe and effective, and can ...
Ted Feldman, M.D., MSCAI FACC FESC, director of the cardiac cath lab, Evanston Hospital, explains the current state of ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
November 7, 2017 — Medtronic plc recently presented positive data for its self-expanding Intrepid transcatheter mitral ...
November 7, 2017 — A new study has found that a pioneering device to repair heart valves is safe and effective, and can ...
November 6, 2017 — In the Hybrid REvascularization Versus Standards (HREVS) study that evaluated three different ...


 November 09, 2017
November 09, 2017