Farouc Jaffer, M.D., Ph.D., director of coronary interventions at Massachusetts General Hospital, discusses the newest ...
September 26, 2017 — The one-year results of the SENTRY clinical trial were presented by principal investigator Michael ...
September 26, 2017 — Treatment of heart attack patients depends on their history of cancer, according to research ...
September 20, 2017 — Shockwave Medical reported positive results from the DISRUPT BTK Study, which were presented at the ...
September 19, 2017 — ScImage Inc. and Invia Imaging Solutions recently announced formation of a joint partnership at the ...

Cardiac nuclear myocardial perfusion imaging (MPI) has been a mature area of imaging for years, but has recently started ...
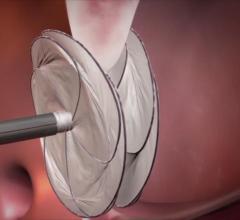
September 18, 2017 — Philips announced the two-year results from the ILLUMENATE European randomized clinical trial (EU ...
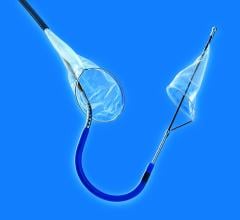
September 18, 2017 – Claret Medical announced publication of a new study in the Journal of the American College of ...
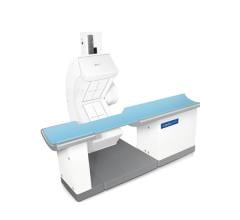
September 15, 2017 — Philips highlighted its newest solution for nuclear cardiology, CardioMD IV, at the American ...
September 14, 2017 — Marijuana use is associated with a three-fold risk of death from hypertension, according to ...

September 14, 2017 — Here are quick summaries for all the key late-breaking vascular and endovascular clinical trials ...
Ziyad Hijazi, M.D., MPH, MSCAI, FACC, director of the cardiac program and chair of the Department of Pediatrics at Sidra ...
September 13, 2017 — Philips announced its presence at the Vascular Interventional Advances (VIVA 17) Annual Conference ...
September 13, 2017 — A subset analysis of the DETOUR I clinical trial showed promising safety and effectiveness results ...
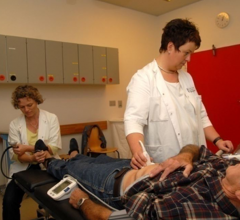
September 12, 2017 — A new screening program for vascular disease saves one life for every 169 men assessed, according ...


 September 28, 2017
September 28, 2017