November 8, 2017 — Investigators in the FAVOR II China and FAVOR II Europe-Japan studies recently presented their ...
November 8, 2017 — Medtronic plc recently presented new clinical research to support the positive clinical performance ...
Philippe Genereux, M.D., co-director of the structural heart program at the Gagnon Cardiovascular Institute at ...
Stephen Worthley, MB, BS, Ph.D., director of cardiac catheterization, University of Adelaide, Australia, presents the ...
A discussion with Ajay Kirtane, M.D., SM, director of the cardiac catheterization laboratories at New York-Presbyterian ...

November 7, 2017 – A new study has found that a pioneering device to repair heart valves is safe and effective, and can ...
Ted Feldman, M.D., MSCAI FACC FESC, director of the cardiac cath lab, Evanston Hospital, explains the current state of ...
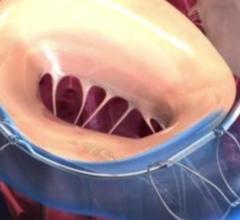
November 7, 2017 — Medtronic plc recently presented positive data for its self-expanding Intrepid transcatheter mitral ...
November 7, 2017 — A new study has found that a pioneering device to repair heart valves is safe and effective, and can ...
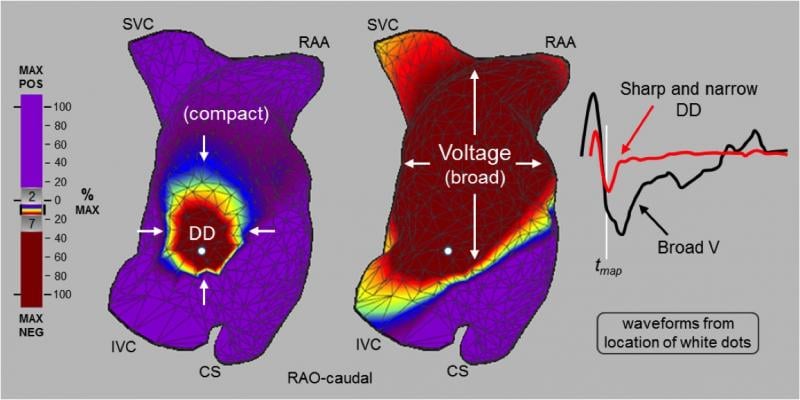
November 7, 2017 — Here is an aggregated list of articles detailing the latest clinical data and new device technology ...
November 6, 2017 — In the Hybrid REvascularization Versus Standards (HREVS) study that evaluated three different ...
November 6, 2017 – Results from ORBITA, a prospective multi-center randomized blinded placebo-controlled study, found no ...
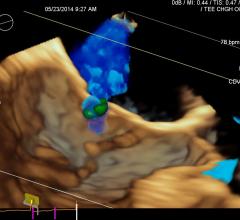
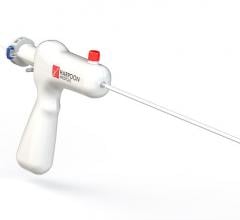
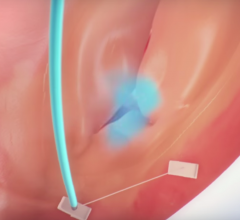
November 6, 2017 — One year follow-up data from the SCOUT I Early Feasibility Study for the Mitralign Trialign System ...
November 6, 2017 — The first trial to evaluate the safety of dual antiplatelet therapy (DAPT) for less than 12 months ...
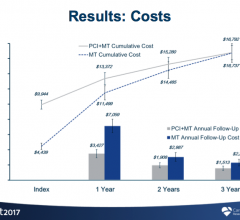
November 6, 2017 — Three-year data from the FAME 2 study show patients with coronary artery disease who underwent a ...

 November 08, 2017
November 08, 2017