July 9, 2024 — Disparities in cardiovascular disease outcomes between urban and rural areas continue to widen, yet ...
Heart Failure
This heart failure channel offers news and new technology to treat heart failure. This includes for new innovations to treat congestive heart failure (CHF). The channel includes news on HFpEF and HFrEF. Heart failure occurs when the heart is no longer able to pump as much blood as the body requires. This can lead to enlargement of the heart because the muscle works harder to supply blood, but the pumping is ineffective. This may be due to defects in the myocardium, such as an infarct, or due to structural issues such as severe valve regurgitation. The disease is divided into four New York Heart Association (NYHA) classes. Stage IV heart failure is when the heart is completely failing and requires a heart transplant or a left ventricular assist device (LVAD).

From FDA approvals to new clinical trials, here is a look at DAIC's top-read content during the month of June:
1. Med ...
July 1, 2024 — CVRx, Inc., a commercial-stage medical device company, announced three additions to its senior leadership ...
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
June 28, 2024 — AstraZeneca has set out a new ambition for Healthy Heart Africa, its flagship health equity initiative ...
June 27, 2024 — The human body has sophisticated defenses against the deposition of calcium minerals that stiffen heart ...
June 27, 2024 — The sheer scale of undiagnosed heart valve disease in our aging population has been revealed for the ...
New research shows superiority of Ultromics’ AI in predicting cardiac-related death As the use of artificial ...
June 27, 2024 — Cardiovascular disease is the leading cause of death for women, and gaps in care and access persist ...
June 24, 2024 — Endotronix, Inc., a privately held company dedicated to advancing the treatment of heart failure (HF) at ...
June 24, 2024 — Aakriti Gupta, MD, and Michelle Kittleson, MD, PhD, cardiologists in the Smidt Heart Institute at Cedars ...
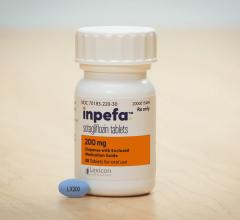
June 21, 2024 — Lexicon Pharmaceuticals, Inc. announced that the peer-reviewed Journal of the American College of ...
June 19, 2024 — When electrophysiologist Eugenio Cingolani, MD, isn’t seeing patients, he can usually be found in his ...
June 19, 2024 — Modern therapies have extended the lives of many cancer patients; however, survivors often live with ...

While the spotlight in 2023 shone on developments around Generative Artificial Intelligence (AI), away from the ...
June 18, 2024 — As we commemorate Juneteenth 2024, JACC, the flagship journal of the American College of Cardiology, has ...
June 14, 2024 — Until now, known risk factors for cardiac arrest have been limited to cardiovascular conditions such as ...

 July 09, 2024
July 09, 2024