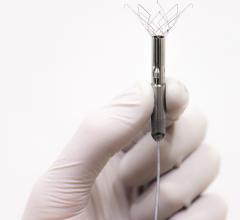
November 2, 2015 — Miracor Medical Systems GmbH announced that its PiCSO system was highlighted during the 2015 ...
Hemodynamic Support Devices
This hemodynamic support systems channel includes content on intra-aortic balloon pumps (IABP), percutaneous ventricular assist devices (pVAD) like the Impella or TandemHeart, extracorporeal membrane oxygenation (ECMO), and ventricular assist devices (VAD). This channel also includes use of these devices in support of patients in cardiogenic shock and advanced heart failure.
October 30, 2015 — Results from the SHIELD I (Coronary InterventionS in HIgh-Risk PatiEnts Using a Novel Percutaneous ...
Manesh Patel, M.D., associate professor of medicine, and director of interventional cardiology and the cath labs at Duke ...
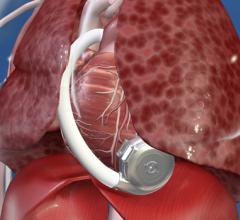
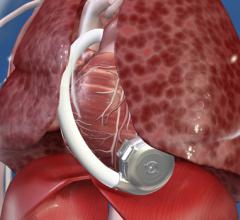
October 28, 2015 — St. Jude Medical Inc. announced the company has received CE Mark approval for the HeartMate 3 Left ...
October 6, 2015 — The novel Aortix device has earned a spot among the diagnostic and therapeutic modalities accepted for ...
October 5, 2015 — Procyrion Inc. of Houston has been awarded the grand prize of $20,000 in the 2015 "Create the Future" ...
September 18, 2015 — SynCardia Systems initiated a Class I recall of the Freedom Driver Systems used with the company’s ...
September 16, 2015 — Thoratec Corp. issued a voluntary Urgent Medical Device Correction Letter to all hospitals who have ...
September 8, 2015 — In a new study reported in The Journal of Thoracic and Cardiovascular Surgery, researchers found a ...
August 11, 2015 — HeartWare International Inc. announced that enrollment in the ENDURANCE2 destination therapy clinical ...
August 7, 2015 — Mayo Clinic is announcing results of a study on the effectiveness of left ventricular assist devices ...
August 6, 2015 — The U.S. Food and Drug Administration (FDA) is alerting healthcare providers, patients and caregivers ...
August 5, 2015 — HeartWare expanded the Class I recall of its ventricular assist device in mid-June due to concerns over ...
August 4, 2015 — Over the past several years, advanced heart failure patients who have not responded to medical therapy ...


 November 02, 2015
November 02, 2015