Hemodynamic Support Devices
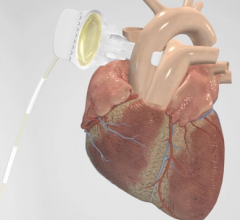
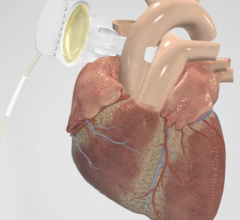
This hemodynamic support systems channel includes content on intra-aortic balloon pumps (IABP), percutaneous ventricular assist devices (pVAD) like the Impella or TandemHeart, extracorporeal membrane oxygenation (ECMO), and ventricular assist devices (VAD). This channel also includes use of these devices in support of patients in cardiogenic shock and advanced heart failure.
December 11, 2013 – The National Cheng Kung University Heart Science and Medical Devices Research Center (NCKU HSDMRC) ...
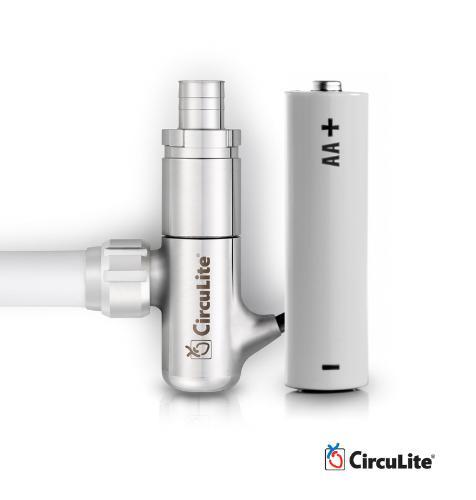
October 1, 2013 — Sunshine Heart Inc. has launched a website dedicated to providing information to patients suffering ...
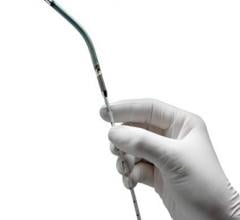
September 19, 2013 — CardiacAssist launched the Protek17 Arterial Cannula, a key component of the TandemHeart temporary ...
There is no shortage of debate among interventional cardiologists over whether vascular closure devices should be used ...
August 20, 2013 — Cardiac surgeons and cardiologists at the University of Maryland Heart Center are part of a multi ...
August 8, 2013 — Sorin Group has received 510(k) clearance for commercialization of Sorin Connect, Sorin Group’s ...
August 5, 2013 — Abiomed Inc. reported that physicians have implanted more than 15,000 Impella pumps in U.S. patients ...

July 19, 2013 — The Mount Sinai Medical Center is the first medical center in New York City to offer the HeartMate II ...
July 11, 2013 — Abiomed’s Impella percutaneous left ventricular assist device (pLVAD) was recently deployed at Baylor ...

June 7, 2013 — Extracorporeal membrane oxygenation (ECMO), a procedure traditionally used during cardiac surgeries and ...
May 20, 2013 — CircuLite Inc. announced that it has received approval from the Federal Agency for Medicines and Health ...
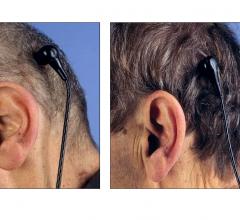
May 16, 2013 — Leviticus Cardio performed a successful animal surgical trial using its wireless coplanar energy transfer ...

 January 20, 2014
January 20, 2014