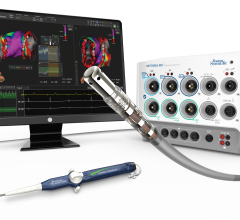
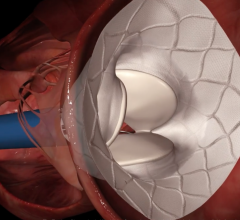
Of patients over age 65 who received an implantable cardioverter-defibrillator (ICD) after surviving sudden cardiac arrest or a near-fatal arrhythmia, almost 80 percent survived two years, according to a new study in the Journal of the American College of Cardiology. This is a higher rate than found in past trials performed to demonstrate the efficacy of the devices in this situation.
Lumedx Corp. recently announced that it has implemented Phase 1 of a long-term, multiphase cardiovascular information system (CVIS) project with Baylor Scott & White Health.
Investigators at the University of Utah have identified distinct differences in the hearts of advanced heart failure patients who have defied the odds and showed signs of recovery from the disease. Published online in the journal Circulation, the new findings could help clinicians identify the best candidates for cardiac recovery therapies.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
The new SunTech Oscar 2 24-hour ambulatory blood pressure monitoring system is now available with the True24 ABPM Patient Diary mobile app, which links to the Oscar 2 via Bluetooth. The True24 app provides a tutorial on ABPM, and prompts the patient to enter information about their activity, posture or any symptoms experienced during a physician-prescribed ABPM study. This diary information helps clinicians interpret the ambulatory blood pressure study data to make well-informed hypertension treatment decisions.
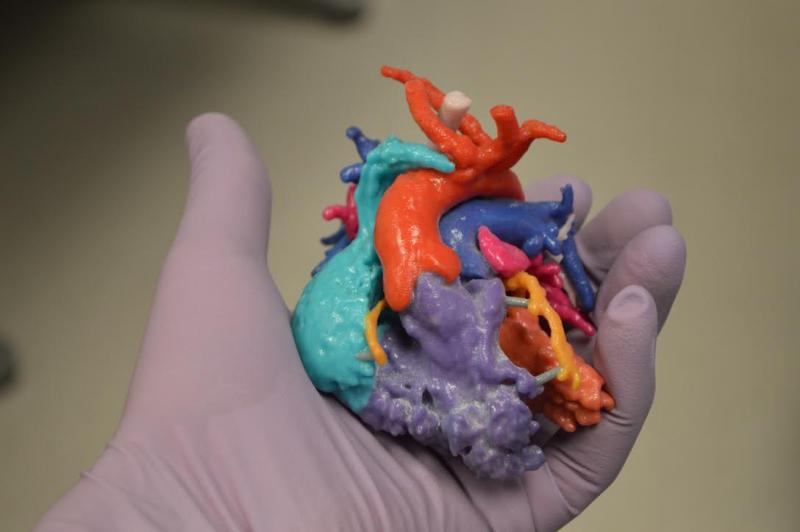
James Laskaris, emerging technology analyst at MD Buyline, recently offered his perspective on new medical technology innovations that might be just months or a few years away.

One of the big advancements in drug-eluting stent (DES) technology has been the development of bioresorbable polymers ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Frost & Sullivan has released a new report, “Vision 2025 – Future of Healthcare,” part of the company’s Advanced Medical Technologies, that identifies 18 technologies that will impact healthcare paradigms by 2025.
January 16, 2017 — BioSig Technologies Inc. announced that the company’s Pure EP System, a novel cardiac ...

Each year imaging system manufacturers use the Radiological Society of North America (RSNA) meeting at the end of the ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
January 16, 2017 – In 2015, researchers and physicians at the Minneapolis Heart Institute Foundation (MHIF) were the ...
BioTrace Medical Inc. announced in December the first commercial use of the company’s Tempo Temporary Pacing Lead since U.S. Food and Drug Administration (FDA) 510(k) clearance in October 2016.
January 13, 2017 — Results from TRANSFORM-OCT, a prospective, randomized trial using optical coherence tomography (OCT) ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
Detailed imaging is needed to support complex interventions for the assessment and procedure guidance. See how the GE ...
Four organizations have announced they are forming a new, multi-stakeholder collaboration, Xcertia, dedicated to improving the quality, safety and effectiveness of mobile health applications. The initial supporters of the collaboration include the American Heart Association (AHA), the American Medical Association (AMA), DHX Group and the Healthcare Information and Management Systems Society (HIMSS).
Acist Medical Systems Inc. announced that enrollment is complete for its Fractional Flow Reserve (FFR) Study – Assessment of Catheter-based Interrogation and Standard Techniques for Fractional Flow Reserve measurement. The study, which began in November 2015, included 240 patients at 12 sites across the United States. Final results are expected to be released in mid-2017.


 January 17, 2017
January 17, 2017