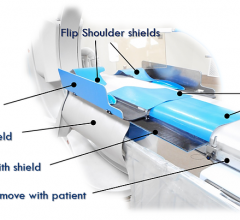
Avinger Inc. announced that the company received 510(k) clearance from the U.S. Food & Drug…
Guerbet LLC USA announced the upcoming launch of SeQure and DraKon, two novel microcatheters for…
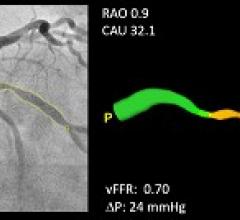
Pie Medical Imaging announced that clinical data on its CAAS vFFR (Cardiovascular Angiographic…
May 21, 2018 — Cardiology medical device reprocessing company Innovative Health recently…
Itamar Medical Ltd. announced the launch of SleePath, the first integrated e-health sleep apnea…
EnvoyAI announced a new integration with Insignia Medical Systems’ InSight PACS (picture…
We started our design from the ground up, tested many prototypes, did not want to compromise on…
Infinitt North America announced that Infinitt Cardiology Suite-v 1.0.8.1 has achieved…
May 9, 2018 — The U.S.
Biosense Webster Inc. announced its Carto Vizigo Bi-directional Guiding Sheath is now available…
The U.S. Food and Drug Administration (FDA) has qualified the Minnesota Living with Heart…
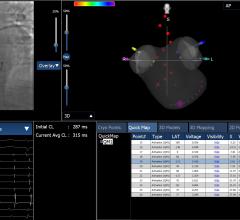
May 3, 2018 — APN Health LLC announced its Navik 3D…
May 3, 2018 — The U.S.
May 3, 2018 — The U.S.
May 2, 2018 — Conavi Medical Inc. has received 510(k) clearance from the U.S.
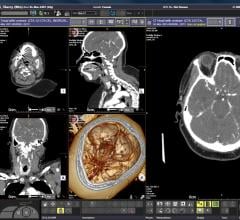
Cerebrovascular imaging analysis company iSchemaView received final clearance from the U.S. Food…
Point of Care Decision Support (PCDS) announced the release of a new version 2.0 of their…
Physio-Control, now part of Stryker, announced its newest version of the LUCAS 3 Chest…
April 18, 2018 — The U.S.
April 17, 2018 — The U.S.


 May 24, 2018
May 24, 2018