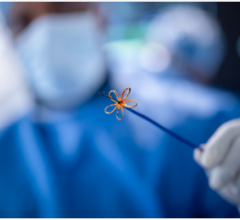
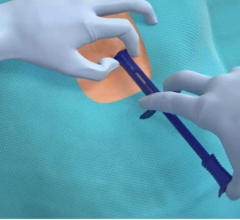
July 21, 2025. — Electrophysiologists at the Texas Cardiac Arrhythmia Institute (TCAI) at St. David's Medical Center ...
EP Lab
This channel includes news and new technology innovations for cardiac electrophysiology (EP) systems, techniques and devices using in EP labs. This includes implantable EP devices, pacemakers, implantable cardioverter defibrillators (ICD), cardiac resychronization therapy (CRT), ablation technologies, left atrial appendage (LAA) occlusion, atrial fibrilation (AF) and Holter monitors.
July 1, 2025 — Stereotaxis, a provider of surgical robotics for minimally invasive endovascular intervention, has ...
July 7, 2025 — Boston Scientific Corp. has received U.S. Food and Drug Administration (FDA) approval to expand the ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
Every second counts when someone is having a stroke and immediate care can mean the difference between life and death ...
June 13, 2025 — An international study has shown that targeted online education on atrial fibrillation (AF) for health ...
May 22, 2025 — Auxira Health has officially launched — the company provides a new approach to clinical support purpose ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
May 21, 2025 — Corify Care recently announced it has entered into a know-how agreement with Mayo Clinic. Corify Care is ...
May 15, 2025 – Royal Philips, a global leader in health technology, has released its 10th annual Future Health Index ...
The American Heart Association recently announced that Dr. Christine E. Seidman, an internationally renowned physician ...
Washington Health System (WHS) provides healthcare services at more than 40 offsite locations across three counties in ...
April 22, 2025 — Orchestra BioMed Holdings, a biomedical company accelerating high-impact technologies to patients ...
April 25, 2025 – Medtronic plc, a global leader in healthcare technology, recently received U.S. Food and Drug ...
April 27, 2025 – The Heart Rhythm Society (HRS) has announced the findings of new studies demonstrating the safety and ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
April 25, 2025 — The annual Society for Cardiovascular Angiography & Interventions (SCAI) meeting, SCAI 2025 Scientific ...
April 25, 2025 – Findings from two late-breaking clinical trials demonstrate the safety and efficacy of left bundle ...
April 24, 2025 – The Heart Rhythm Society (HRS) has unveiled new research that underscores the critical role of ...


 July 21, 2025
July 21, 2025