Dec. 3, 2025 — Recross Cardio Inc., a structural heart company developing next-generation membrane sealing technology ...
EP Lab
This channel includes news and new technology innovations for cardiac electrophysiology (EP) systems, techniques and devices using in EP labs. This includes implantable EP devices, pacemakers, implantable cardioverter defibrillators (ICD), cardiac resychronization therapy (CRT), ablation technologies, left atrial appendage (LAA) occlusion, atrial fibrilation (AF) and Holter monitors.
Nov. 10, 2025 — Stereotaxis has received U.S. Food and Drug Administration 510(k) clearance for its latest generation ...
Oct. 22, 2025 -- Medical robotics innovator Noah Medical presented two new data sets highlighting the clinical and ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The newer ECG devices we have now are so much less cumbersome. It’s like wearing a Band-Aid versus carrying a bulky device,” said the Greenwood, Mississippi internist. “My patients prefer the more comfortable, wire-free form factor, and the quality is as good as, or better, than the Holter,” continued Boler. “Plus, my patient compliance has increased. With the Holter, the leads sometimes come off. The patient may think the device isn’t working, so they take it off and we have to restart the process.”
Oct. 10, 2025 — Johnson & Johnson MedTech, in collaboration with the Heart Rhythm Clinical and Research Solutions, LLC ...
Sept. 23, 2025 — CeleCor Therapeutics’ multinational Phase 3 clinical trial of its investigational heart-attack drug ...
Sept. 22, 2025 — The latest findings on heart failure (HF) published by the Heart Failure Society of America (HFSA) reve ...
Sudden cardiac death (SCD) is the leading medical cause of death in young athletes and its impact is consistent worldwide. Most professional athletes in the United States are required to take part in comprehensive cardiovascular screening programs to identify often-asymptomatic congenital or inherited heart disorders, and other cardiac risk factors. There remains a debate however, whether to mandate ECGs as part of pre-participation screening programs for student athletes at the collegiate and high school levels or even at younger ages.
Sept. 8, 2025 — AMO Pharma Ltd., a clinical-stage specialty biopharmaceutical company focusing on rare genetic ...
Sept. 2, 2025 — Johnson & Johnson MedTech has announced acute safety and effectiveness results from the Varipure ...
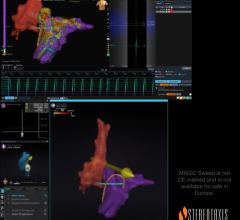
Sept. 2, 2025 — Stereotaxis has announced the successful completion of the world’s first procedures using MAGiC Sweep, a ...
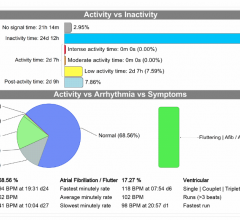
Rapid advances in technology to monitor atrial fibrillation (AF or Afib) are enabling clinicians to access real-time ...

Patients with type 2 diabetes (T2D) and chronic kidney disease (CKD) are at elevated risk for cardiovascular disease.1 Y ...
Sept. 3, 2025 — Kardium Inc. recently announced it has received pre-market approval (PMA) for the Globe Pulsed Field ...
Aug. 26, 2025 — Octane, an innovative organization building the SoCal of Tomorrow by connecting people, resources and ...
There is a movement toward real-time, remote cardiac monitoring with the latest generation of event and Holter monitor ...
Aug. 07, 2025 —- New research by European cybersecurity company Modat revealed more than 1.2 million internet-connected ...
July 28, 2025 — Stereotaxis, a provider of surgical robotics for minimally invasive endovascular intervention, has ...
July 25, 2025 — Data in recent staffing surveys from the American Society of Radiologic Technologists show that vacancy ...


 December 04, 2025
December 04, 2025