April 9, 2026 — Mount Sinai researchers have created an analytic tool using machine learning that can predict ...
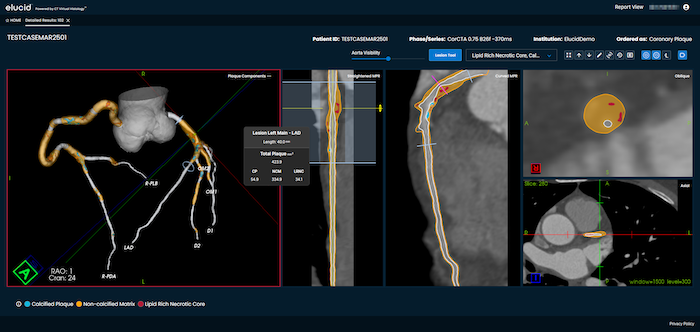
For decades, cardiology has evaluated coronary artery disease (CAD) largely through the lens of luminal stenosis, or the ...
April 13, 2026 — Johnson & Johnson has announced favorable 12-month interim effectiveness and safety results from ...
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
April 13, 2026 — Penumbra, Inc. announced 90-day results of the landmark STORM-PE randomized controlled trial (RCT) ...
April 13, 2026 —The American Heart Association (AHA) has granted Case Western Reserve University the Rapid Impact ...
April 9, 2026 — Cardiovascular demand continues to rise across the United States, placing increasing pressure on ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
April 7, 2026 —TRiCares SAS has received approval from the U.S. Food and Drug Administration (FDA) for an ...
April 8, 2026 — Anumana, Inc. recently announced U.S. Food and Drug Administration (FDA) clearance of its ECG-AI ...
April 6, 2026 —Stereotaxis has received U.S. Food and Drug Administration 510(k) clearance for its Synchrony system ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
April 7, 2026 — BD (Becton, Dickinson and Co.) has launched the HemoSphere Stream Module, a new innovation which gives ...
March 28, 2026 — Amgen announced today that Repatha (evolocumab), when added to statins or other low-density lipoprotein ...

Four vital signs make up the traditional core of clinical diagnosis: heart rate, blood pressure, respiratory rate and ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
March 31, 2026 — Solo Pace Inc. has announced the commercial release of the SoloPace Fusion Temporary Pacing System. The ...
April 2, 2026 — Omron Healthcare Co. has launched a randomized controlled trial in collaboration with the University of ...
April 2, 2026 — Iterative Health and US Heart & Vascular (USHV) have announced a strategic partnership to advance ...


 April 14, 2026
April 14, 2026
















