
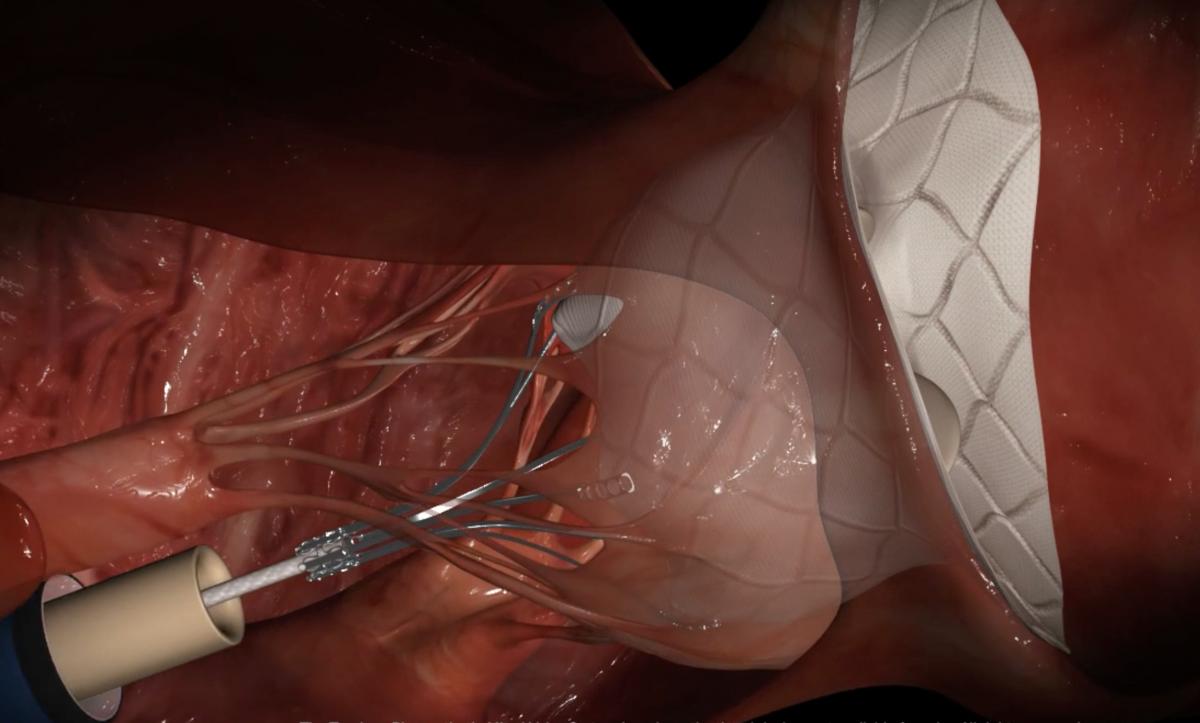
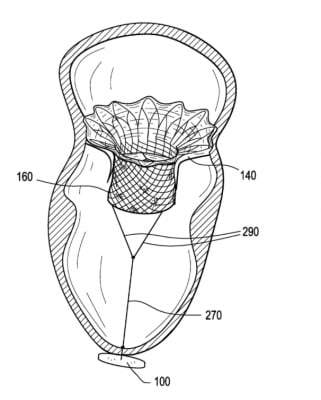
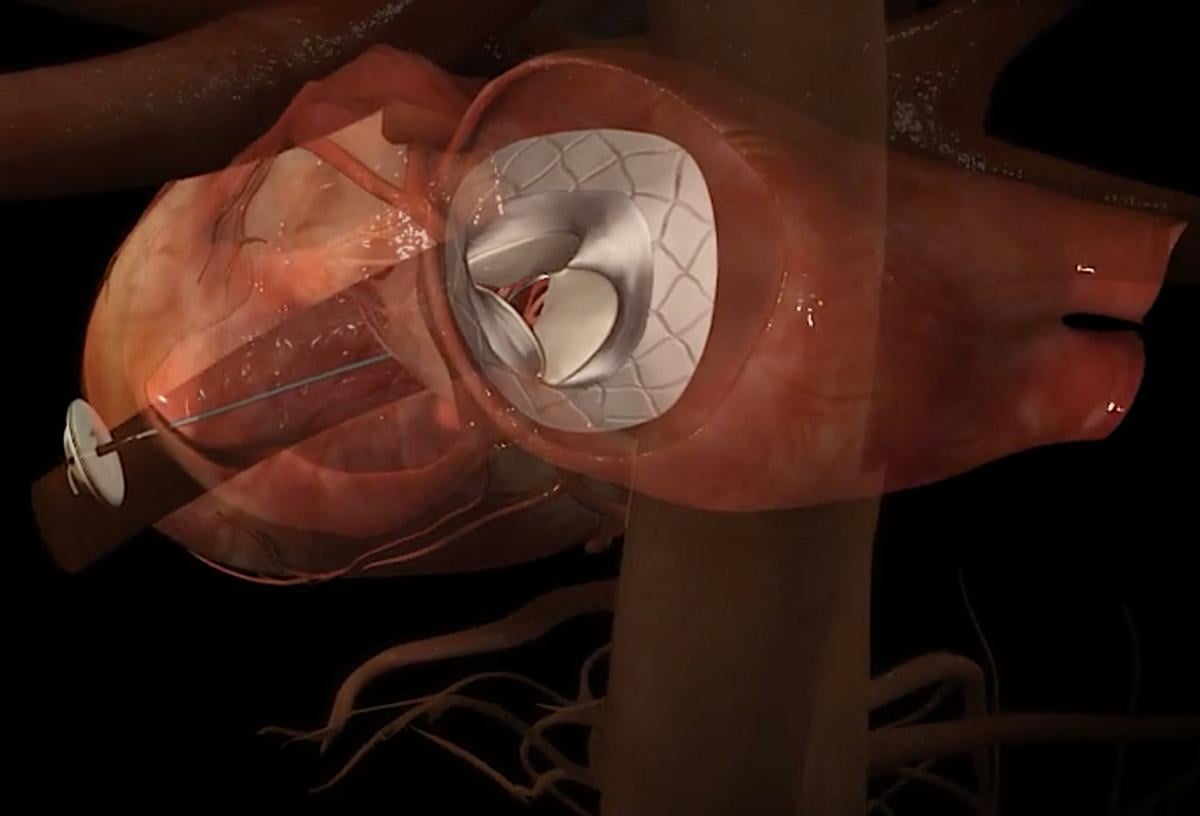
The Abbott Tendyne transcatheter mitral valve replacement (TMVR) system uses an anchor attached to the apex of the heart with a whether line attached to the valve. This helps to keep the valve anchored in the mitral annulus and prevent embolization. This anchor system was used because, unlike the aortic valve, the mitral valve has a very thin landing zone to secure the valve. The design also eliminated any hardware hanging or folding into the left ventricle, which could cause left ventricular outflow track (LVOT) obstruction, which can prevent blood flow through the aortic valve and to the rest of the body.
January 30, 2020 — The Abbott Tendyne Transcatheter Mitral Valve Implantation (TMVI) system received European CE mark this week. It is the first transcatheter mitral valve replacement (TMVR) technology to gain commercial clearance in the world. It addresses a critical need to eliminate mitral regurgitation when surgery or mitral repair is not an option in high-risk surgical patients.
This therapy treats significant mitral regurgitation (MR) in patients requiring a heart valve replacement. It provides a safe and effective solution for MR patients who are not candidates for open-heart surgery or transcatheter mitral valve repair.
 As the most common heart disease affecting the heart valves, MR is a debilitating, progressive and life-threatening condition in which the heart's mitral valve does not close completely, allowing blood to flow backward into the left atrium of the heart. Due to the complex nature of mitral valve disease and different patient anatomies, multiple treatment options for MR may be necessary. For patients at high-risk for open-heart surgery or in clinical situations when the mitral valve is too damaged for a successful repair with Abbott's MitraClip device, the Tendyne system offers an alternative minimally invasive treatment option.
As the most common heart disease affecting the heart valves, MR is a debilitating, progressive and life-threatening condition in which the heart's mitral valve does not close completely, allowing blood to flow backward into the left atrium of the heart. Due to the complex nature of mitral valve disease and different patient anatomies, multiple treatment options for MR may be necessary. For patients at high-risk for open-heart surgery or in clinical situations when the mitral valve is too damaged for a successful repair with Abbott's MitraClip device, the Tendyne system offers an alternative minimally invasive treatment option.
The Abbott Tendyne valve is a first-of-its-kind therapy to replace the mitral valve in patients in need of symptom relief and quality-of-life improvement without open surgery and when transcatheter mitral repair is not possible. Global trial results to date have demonstrated excellent procedural safety and have shown 98.9 percent of Tendyne patients experienced MR elimination at discharge which was sustained through one-year in this very sick patient group.[1-5]
 "European approval for Abbott's Tendyne mitral valve replacement therapy provides the clinical community with a new choice in how we approach correcting a leaking mitral valve," said Hendrik Treede, M.D., professor of cardiac surgery, University Hospital Bonn, Germany. "For the first time outside of clinical trial settings, heart teams now have a minimally invasive valve replacement therapy that is backed by an excellent safety profile and designed to help physicians reposition the device as needed for improved patient outcomes."
"European approval for Abbott's Tendyne mitral valve replacement therapy provides the clinical community with a new choice in how we approach correcting a leaking mitral valve," said Hendrik Treede, M.D., professor of cardiac surgery, University Hospital Bonn, Germany. "For the first time outside of clinical trial settings, heart teams now have a minimally invasive valve replacement therapy that is backed by an excellent safety profile and designed to help physicians reposition the device as needed for improved patient outcomes."
A post-approval study of the therapy will also be co-led by Nicolas Dumonteil, M.D., interventional cardiologist, Clinique Pasteur, France.
The Tendyne TMVI system can adapt to a range of patient anatomies thanks to its innovative and unique design. The self-expanding valve is delivered through a small incision in the chest and up through the heart where it is implanted in a beating heart, replacing the person's native mitral valve. The Tendyne valve is available in multiple sizes to treat a broad range of valve anatomies. The valve is fully repositionable and retrievable during implantation, allowing for the best possible outcome for people suffering from MR and needing a valve replacement.
 "The launch of the Tendyne device builds upon our history of developing groundbreaking therapies that offer new treatment options for people with serious structural heart conditions who have limited treatment options," said Michael Dale, senior vice president of Abbott's structural heart business. "The availability of Tendyne as a treatment option in Europe provides physicians with an additional tool that has been shown to completely correct MR in very ill patients, and it adds to Abbott's portfolio of life-changing and life-saving treatments."
"The launch of the Tendyne device builds upon our history of developing groundbreaking therapies that offer new treatment options for people with serious structural heart conditions who have limited treatment options," said Michael Dale, senior vice president of Abbott's structural heart business. "The availability of Tendyne as a treatment option in Europe provides physicians with an additional tool that has been shown to completely correct MR in very ill patients, and it adds to Abbott's portfolio of life-changing and life-saving treatments."
Abbott is developing transcatheter mitral valve technologies as alternatives to open-heart surgery. Building upon its success with the MitraClip device and many years of mitral valve experience and clinical evidence, Abbott is leading the way in novel, transcatheter devices by investing in the development and clinical study of minimally invasive technologies.
SUMMIT Pivitol U.S. Clinical Study Evaluating Tendyne in the U.S.
The Tendyne system is an investigational device in the United States. In July 2018, Abbott initiated the Treatment of Symptomatic Mitral Regurgitation (SUMMIT) clinical study in the U.S. for the TMVR system. Abbott will use the SUMMIT data for a market clearance submission to the U.S. Food and Drug Administration (FDA). The Tendyne device allows repositioning and retrieval, enabling more accurate device placement during implantation for better outcomes. The trial is enrolling up to 1,010 patients at 80 sites in the U.S., Europe and Canada.
Related Tendyne Transcatheter Mitral Calve Replacement Content:
Abbott Begins Tendyne Transcatheter Mitral Valve U.S. Pivotal Trial
Developments in Transcatheter Mitral Valve Replacement
First Tendyne Transcatheter Mitral Valve Implanted in New York
Tendyne Transcatheter Mitral Valve Replacement Device Demonstrates Positive 30-Day Outcomes
Minneapolis Heart Institute Continues Early Feasibility Trial With Tendyne Device for Smaller Hearts
Advances and Future Directions for Transcatheter Valves
Abbott Expands Mitral Valve Therapy Offerings With Pair of Transactions
Minneapolis Heart Institute Foundation Conducts First U.S. Implant of TMVR Device
Transcatheter Mitral Valve Replacement Devices in Development
References:
3. Expanded Clinical Study of the Tendyne Mitral Valve System—Global Feasibility Study (NCT02321514); still recruiting. https://clinicaltrials.gov/ct2/show/NCT02321514, Jan. 30, 2020.
5. Feasibility Study of the Tendyne Mitral Valve System for Use in Subjects With Mitral Annular Calcification (NCT03539458); No longer recruiting. Study includes Emory University Hospital, Atlanta; Kansas Cardiovascular Research Institute of Kansas, Wichita; Minnesota Abbott Northwestern Hospital; and West Virginia University, Morgantown. https://clinicaltrials.gov/ct2/show/NCT03539458. Accessed Jan. 30, 2020.


 May 13, 2026
May 13, 2026 









