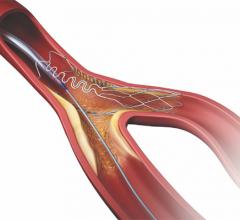
July 26, 2017 — Intact Vascular Inc. recently announced that its Tack Optimized Balloon Angioplasty II Below the Knee ...
Stents
This channel includes news and new technology innovations for stents, also called vascular scaffolds. Stents are used to help prop open a vessel treated by balloon angioplasty because of the barotrauma caused by the extreme stretching of vessel walls. The stent enables to vessel to heal in an open position with collapsing. Drug eluting stents (DES) are coated in anti-proliferative drugs to precent scar tissue growth which can cause restenosis and occlude the vessel. DES require antiplatelet therapy because the drug carrier polymer on DES can cause thrombus inside the stent, even years after treatment, which is why bare metal stents are still used in some patients. This page includes news on coronary stents, carotid stents, peripheral stents, bioresorbable stents, and renal stents.
July 17, 2017 — LimFlow SA announced enrollment of the first patient in the U.S. feasibility study of the LimFlow ...
July 14, 2017 — Intact Vascular Inc. announced the U.S. Food and Drug Administration (FDA) approved an Investigational ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
June 12, 2017 – The Medtronic Resolute Onyx Drug-Eluting Stent (DES) met its primary endpoint of target lesion failure ...
May 12, 2017 — PinnacleHealth CardioVascular Institute enrolled the first patient in Pennsylvania in a trial assessing ...
May 1, 2017 — The U.S. Food and Drug Administration (FDA) approved Medtronic’s Resolute Onyx Drug-eluting Stent (DES) ...
This video, provided by Medtronic, demonstrates the Resolute Onyx coronary stent. It was cleared by the FDA and launched ...
April 28, 2017 — Biotronik’s Pulsar-18 bare metal stent (BMS) has yielded high primary patency in a real-world setting ...
March 31, 2017 — Tryton Medical Inc. recently announced that the first U.S. commercial case using the Tryton Side Branch ...
Stephen Ellis, M.D., professor of medicine and director of interventional cardiology at Cleveland Clinic, discusses the ...
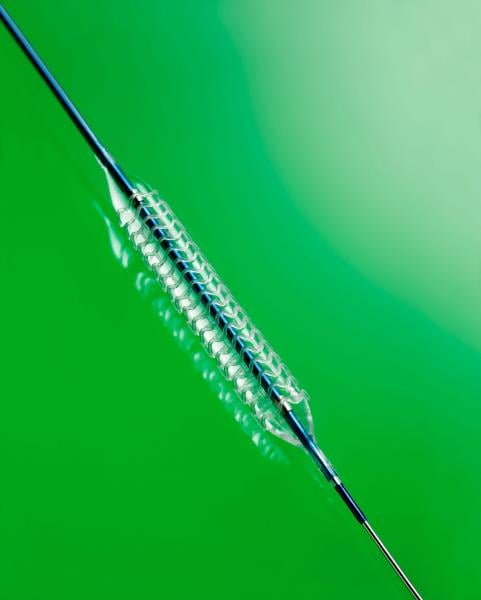
March 21, 2017 — In the late-breaking ABSORB III Trial two year results presented at the American College of Cardiology ...
March 16, 2017 — W. L. Gore & Associates Inc. recently announced the Health Canada approval of the Gore Tigris Vascular ...
This video, provided by Tryton, demonstrates the implantation of the Tryton Side Branch Stent. It became the first ...
March 6, 2017 – The U.S. Food and Drug Administration (FDA) has granted pre-market approval (PMA) for the Tryton Side ...
March 3, 2017 — Biosensors International Group Ltd. announced in February enrollment of the first patient in LEADERS ...

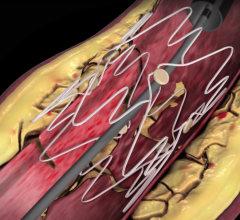
 July 26, 2017
July 26, 2017