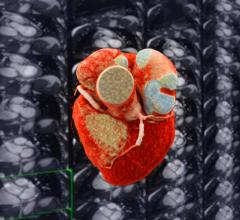
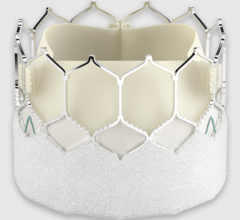
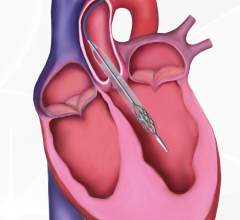
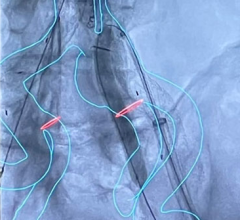
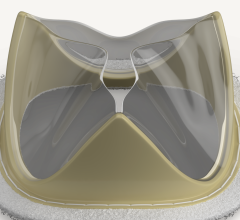
October 19, 2023 — Edwards Lifesciences Corporation announced the company's EVOQUE tricuspid valve replacement system ...
Structural Heart
This structural heart channel includes news, videos, podcasts and other content related to diagnosis and treatment of structural heart disease. Topics covered include heart valve repair and replacement, transcatheter aortic valve replacement (TAVR), transcatheter mitral valve replacement (TMVR), transcatheter tricuspid valve replacement (TTVR), left atrial appendage (LAA) occlusion, heart failure interventional device therapies, and closing holes in the heart using, including occlusion of atrial septal defects (ASDs), ventricular septal defects (VSDs) and patent foramen ovales (PFOs).
October 17, 2023 — The Patel Children's Heart Institute at St. Joseph's Children's Hospital achieved a milestone ...
October 12, 2023 — Franklin Mountain Medical announced today that the Dib UltraNav Transseptal Catheter System, which ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
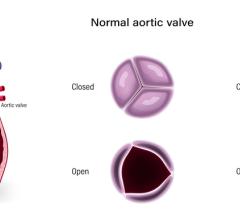
October 5, 2023 — Mayo Clinic researchers have developed a calculation that can help identify moderate aortic stenosis p ...
October 5, 2023 — A newly-issued "Scientific Statement on Durable Mechanical Circulatory Support" in the October issue ...
September 20, 2023 — The Minneapolis Heart Institute Foundation (MHIF), an internationally renowned cardiovascular ...
September 7, 2023 — Boston Scientific Corporation announced it has received U.S. Food and Drug Administration approval ...
September 6, 2023 — The diagnosed severity of aortic stenosis strongly correlates with clinical outcomes, new Kaiser ...
September 6, 2023 — Magenta Medical, developer of the Elevate heart pump system, an FDA Designated Breakthrough Device ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...

Here is a look at what DAIC viewers were reading during the month of July:
July 27, 2023 — Abiomed is recalling the Impella Intravascular Left Sided Blood Pumps because the pump’s Instructions ...


 October 19, 2023
October 19, 2023