April 9, 2024 — Administering tranexamic acid (TxA), a drug used to reduce bleeding during heart surgery, topically ...
Structural Heart
This structural heart channel includes news, videos, podcasts and other content related to diagnosis and treatment of structural heart disease. Topics covered include heart valve repair and replacement, transcatheter aortic valve replacement (TAVR), transcatheter mitral valve replacement (TMVR), transcatheter tricuspid valve replacement (TTVR), left atrial appendage (LAA) occlusion, heart failure interventional device therapies, and closing holes in the heart using, including occlusion of atrial septal defects (ASDs), ventricular septal defects (VSDs) and patent foramen ovales (PFOs).
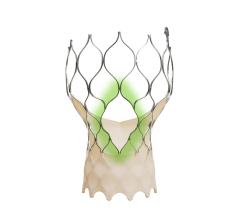
April 9, 2024 — People with a small aortic annulus, a part of the heart’s anatomy where the left ventricle meets the ...
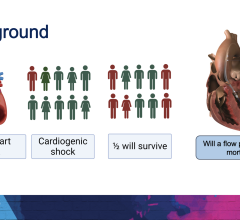
April 8, 2024 — Implantation of the Impella CP micro-axial flow pump in the hours after a heart attack significantly ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
April 4, 2024 — Osso VR, a leader in immersive procedural training, announces the release of a co-developed curriculum ...

Here is a Top 10 look at the content that was trending during the month of March on dicardiology.com:
1. CLS Health ...
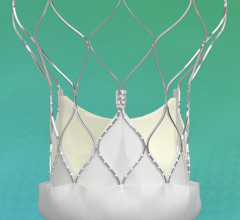
April 2, 2024 — Abbott announced that the U.S. Food and Drug Administration (FDA) approved the company's first-of-its ...
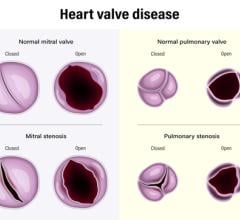
April 1, 2024 — Roughly 25,000 Americans die each year from valvular heart disease, but researchers from Rutgers Health ...
March 28, 2024 — Medtronic plc, a global leader in healthcare technology, announced that the United States Food and Drug ...
March 25, 2024 — In a groundbreaking medical advancement, three esteemed cardiologists from CLS Health, Dr. Bahaeddin ...
March 21, 2024 — The U.S. Food and Drug Administration (FDA) announced that Abiomed is recalling the Instructions for ...
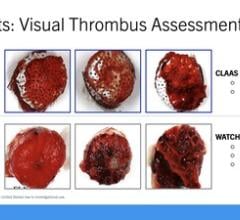
March 18, 2024 — Conformal Medical, Inc. announced that the CLAAS System was featured in a podium presentation at the Ca ...
March 12, 2024 — Medtronic plc, a global leader in healthcare technology, today announced two late-breaking data ...

February was a short but busy month in the diagnostic and interventional cardiology world, with a lot of news being ...
February 26, 2024 — Hackensack Meridian Jersey Shore University Medical Center and Hackensack University Medical Center ...


 April 09, 2024
April 09, 2024